Learning objectives
- Difference between intra-abdominal pressure (IAP) and abdominal compartment syndrome (ACS)
- Recognize ACS and the pathophysiological consequences of ACS
- Management of ACS
Definition and mechanisms
- Normal intra-abdominal pressure (IAP) ranges between 0-5 mmHg while in critically-ill patients, an IAP of 5-7 mmHg is considered normal
- Intra-abdominal hypertension is defined as a sustained intra-abdominal pressure (IAP) ≥12 mmHg
- Abdominal compartment syndrome (ACS) is defined as IAP rises > 20 mmHg thereby leading to new organ dysfunction
- Abdominal perfusion pressure (APP) is calculated as the mean arterial pressure (MAP) minus the IAP
- A critically ill patient with high mortality & morbidity
Signs and symptoms
- Malaise
- Weakness
- Dyspnea
- Abdominal bloating
- Abdominal pain
- Hypoxia
- Hypercabia
- Oliguria
Etiology
Acute ACS
- Primary: due to injury or disease in the abdominopelvic region (e.g., pancreatitis, abdominal trauma)
- Secondary: does not originate in the abdomen or pelvis (e.g., fluid resuscitation, sepsis, burns)
Chronic ACS
- In association with peritoneal dialysis or chronic ascites
Artificially raised IAP
- External compression, for example, prolonged prone positioning for spinal surgery with insufficient provision for abdominal movement
Risk factors
| Diminished abdominal wall compliance | Acute respiratory failure, especially with elevated intrathoracic pressure Abdominal surgery with subjectively tight primary closure Major trauma/burns Prone positioning, head of bed elevated > 30° High BMI, central obesity |
| Increased intra-luminal contents | Gastroparesis Ileus Colonic pseudo-obstruction |
| Increased abdominal contents | Hemoperitoneum/pneumoperitoneum Ascites/liver dysfunction |
| Capillary leak/fluid resuscitation | Metabolic acidosis ( pH < 7.2) Hypotension Perioperative hypothermia Polytransfusion (>10 units of blood/24 h) Coagulopathy (platelets < 55 000 mm-3, prothrombin time > 15 s, partial thromboplastin time > 2 times normal, or international standardized ratio >1.5) Massive fluid resuscitation (> 5 litre/24 h) Pancreatitis Oliguria Sepsis Trauma Burns Damage control laparotomy |
Pathophysiological effects of raised IAP
| Central nervous system | Increased intra-cranial pressure |
| Cardiovascular system | Increased systemic vascular resistance Pulmonary vascular resistance Decreased venous return with concomitant venous congestion |
| Gastrointestinal and hepatic system | Decreased coeliac, mesenteric, hepatic and hepatic portal blood flow Increased oedema, bacterial translocation and liver dysfunction |
| Renal system | Increased renal tubular pressure and urinary obstruction Decreased renal blood flow and urine output |
| Respiratory system | Increased ventilation-perfusion mismatch, ventilatory pressure, basal atelectasis and PaCO2 Decreased chest wall and pulmonary compliance and PaO2 |
Diagnosis
- Indirect measurement of IAP using intragastric, intracolonic, intravesical (bladder), or inferior vena cava catheters
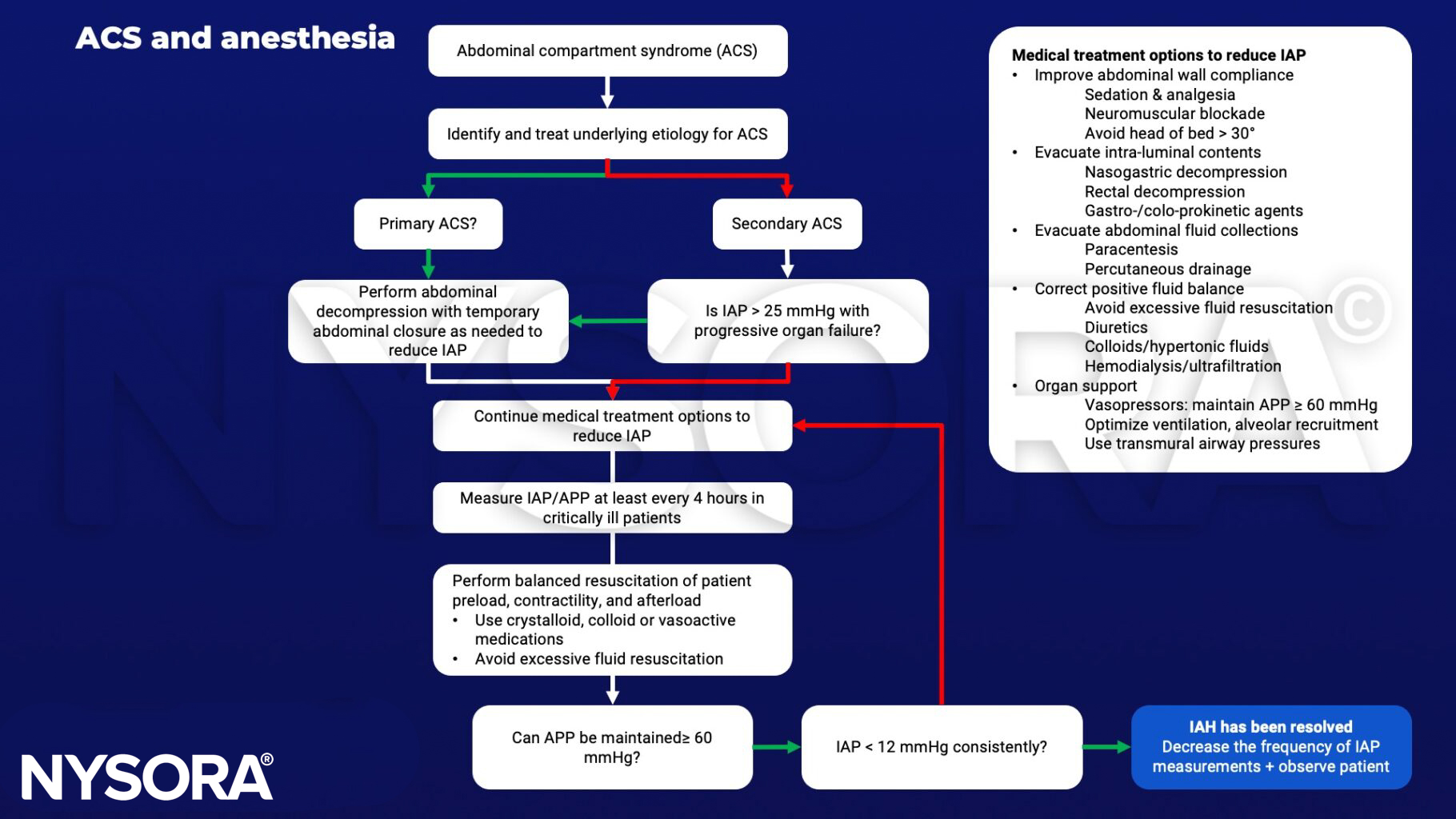
Management
- Patients with two or more risk factors should have IAP monitoring
- Treatment:
- Regimens lowering IAP
- Open the abdominal wound and perform a temporary closure of the abdominal wall with mesh or a plastic bag (Bogota bag)
- Regiments aiming at organ support
- Keep abdominal perfusion pressure (APP) (systemic blood pressure – intra-abdominal pressure) > 60mmHg
Keep in mind
- Consequences of decompression:
- Sudden ↓ cardiac output & SVR
- Reperfusion: risk of systemic acidosis & hyperkalemia
- Possible fatal arrhythmia & arrest
- Sudden change in respiratory compliance (avoid overventilation)
- Avoid bradycardia (preload is compromised & CO may be heart rate dependent)
- Maintain high preload particularly once decompressed
Suggested reading
- Neil Berry, Simon Fletcher, Abdominal compartment syndrome, Continuing Education in Anaesthesia Critical Care & Pain, Volume 12, Issue 3, June 2012, Pages 110–117.
- Mullens W, Abrahams Z, Skouri HN, et al. Elevated intra-abdominal pressure in acute decompensated heart failure: a potential contributor to worsening renal function? J Am Coll Cardiol. 2008;51(3):300-306.
Clinical updates
Goldstein and Nunn (Critical Care Clinics, 2025) describe the expanding role of point-of-care ultrasound in diagnosing and managing IAH/ACS, detailing correlations between elevated IAP and inferior vena cava morphology, venous Doppler congestion patterns (VExUS), increased renal resistive index, and reduced diaphragmatic thickening fraction. They propose ultrasound-derived models combining renal RI, diaphragmatic metrics, and lactate to predict IAH, positioning POCUS as an adjunct to—but not a replacement for—bladder pressure measurement in early detection and physiologic monitoring.
Kumar et al. (Journal of Anaesthesiology Clinical Pharmacology, 2025) report a 14-year-old undergoing percutaneous nephrolithotomy who developed rising airway pressures, hypoxemia, and decreased left-sided air entry in the prone position, with point-of-care ultrasound revealing massive retroperitoneal fluid collection in Morison’s pouch and pleural effusion due to irrigation fluid extravasation. Measured intra-abdominal pressure confirmed acute compartment syndrome, and prompt drainage guided by ultrasound led to hemodynamic and ventilatory improvement, highlighting POCUS as a rapid bedside diagnostic tool for life-threatening ACS during PCNL when clinical assessment is limited.
Fu and Luo (Clinical Case Reports, 2024) describe a 2-month-old infant with abdominal compartment syndrome who developed immediate ventilation failure, hypoxemia, and hemodynamic collapse after induction and endotracheal intubation for emergent laparotomy, unresponsive to deepening anesthesia, muscle relaxation, and epinephrine. Ventilation and circulation were restored only after rapid surgical abdominal decompression, highlighting that in severe pediatric ACS, induction and neuromuscular blockade can precipitate critical increases in intrathoracic pressure, and that surgical decompression must be immediately available when anesthesia is initiated.
Jang et al. (Acute and Critical Care, 2023) review contemporary definitions and management of ACS in ICU patients, emphasizing that sustained IAP > 20 mmHg with new organ dysfunction defines ACS and that more than 50% of ICU patients develop IAH within the first week, with mortality reaching 38.6% when ACS occurs. They advocate risk-factor–based IAP monitoring (every 6–8 h when ≥1 risk factor), early nonsurgical strategies including nasogastric decompression, percutaneous drainage, fluid optimization guided by ultrasound, and timely decompressive laparotomy when medical therapy fails, while cautioning against reliance on abdominal perfusion pressure without stronger interventional evidence.
De Waele (Current Opinion in Critical Care, 2022) highlights the evolving epidemiology and management of intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS), noting a 2.57-fold increase in the risk of acute kidney injury in IAH and independent associations between combined IAH and hypoxemic respiratory failure and increased 28-day mortality. While novel automated, intragastric, and noninvasive IAP monitoring techniques are emerging, transvesical measurement (20 mL instillation) remains the gold standard, and contemporary management emphasizes early risk stratification, judicious fluid balance, targeted nonoperative measures, and improved open abdomen strategies incorporating negative pressure therapy and mesh-mediated fascial traction to achieve ~80% closure rates.