In the usual work of a physician, knowing how to detect a pleural effusion is a conclusion. The interest of the BLUE-protocol is to specify what to do with this information (redundant here, informative there) and how to link it to a cause. The fast detection of pleural effusions is part of the BLUE-protocol, which simplifies the diagnosis by adding original approaches. This familiar application imagined by Dénier in 1946 and assessed by Joyner in 1967 has for many doctors summarized the interest of thoracic ultrasound. Why to use ultrasound in complement with other tools (physical examination and others). Ultrasound evaluates the volume and the nature of an effusion and indicates the appropriate area for a thoracentesis, far better than radiography. For this application, our 5 MHz microconvex probe is perfect.
1. THE TECHNIQUE OF THE BLUE-PROTOCOL
From the old school and during decades, pleural effusions were detected during abdominal examinations, using abdominal probes and subcostal approaches. This route can mislead (Fig. 1). Our microconvex probe is perfect for direct analysis through the intercostal space. Therefore, new signs adapted to this direct approach will be described.

Fig.1 Pleural effusion and traditional approach. This effusion appears during a transabdominal approach, through the liver (L), in a transversal scan. This does not provide a definite diagnosis with certain lower-lobe consolidations and also does not allow ultrasound- guided thoracentesis. Note that the effusion goes posterior to the inferior vena cava (V), a feature that distinguishes, if necessary, pleural from peritoneal effusion
Pleural effusion collects in dependent areas (principle n°2 – fluid is heavier than air). Any free pleural effusion is therefore in contact with the bed in a supine patient. Rotating the patient laterally is sometimes difficult, and not satisfactory if the effusion moves to inaccessible dependent areas (Fig. 2). Scanning only the accessible, lateral wall will result in a loss of sensitivity. We insert the probe at the PLAPS-point, as far as we can (read again carefully the technique of the PLAPS-point).

Fig.2 PLAPS-point and Earth-sky axis. The lateralization maneuver. Left: the probe explores the lateral zone up to bed level. The bed prevents the probe from scanning further. Note the probe is far from perpendicular to the wall. Using this horizontal axis, the detection of the small effusion ( arrowheads ) is not obtained. Right: the back of the patient has been slightly raised (lateralization maneuver) (or the bed is soft enough for avoiding this maneuver). The probe gains precious centimeters of exploration and is now pointing to the sky, at a PLAPS-point, not far from perpendicular. Minimal effusion or posterior consolidation can be diagnosed. Note that the effusion has slightly moved toward the medial line (the arrows indicate the maximal thickness of the fluid, the circle the medial line), indicating that the maneuver of turning the patient should be minimal (a wider maneuver could result in locating this effusion at the mediastinal wall)
The principle of the PLAPS-point is simple: if only one “shot” is allowed for determining whether there is, or not, a pleural effusion, this location indicates immediately quite all free pleural effusions, either abundant or minute. Ultrasound can perfectly detect millimetric effusions (Fig. 3), provided the probe is applied at the correct spot.
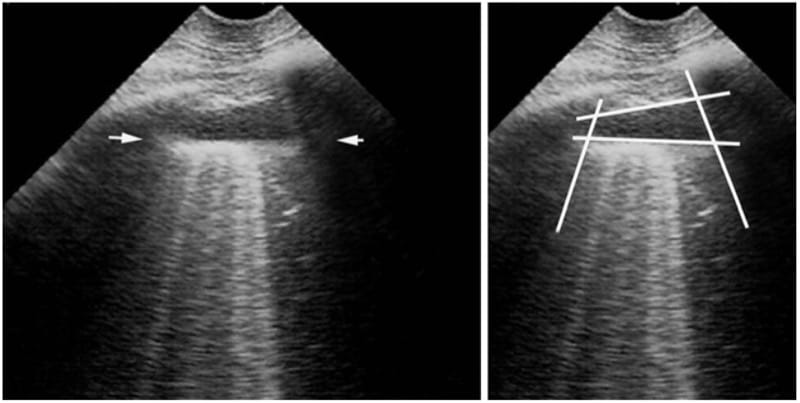
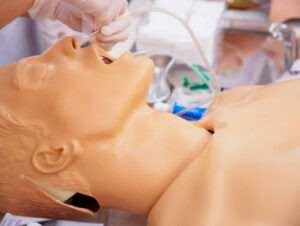
Fig.3 Minimal pleural effusion. Longitudinal scan at the PLAPS-point. This figure indicates several pieces of information.
-
It shows the quad sign: the dark image is an effusion not because it is dark but because it is framed within four regular borders: the pleural line, the shadow of the ribs, and mostly the regular deep border (the lung line – arrows). The quad sign is drawn at the right image.
-
It shows the absence of local lung consolidation, since the image beyond the lung line is artifactual.
-
It indicates the volume of the effusion. The interpleural expiratory distance is 7 mm. This corresponds to a 20–40 ml effusion.
-
This effusion seems too thin for safe thoracentesis.
-
This figure allows to present the sub-B-lines (artifacts looking like B-lines, arising not from the pleural line but from the lung line, whose meaning is not the same, since only the pleural fluid must be on attention). This notion of sub-B-lines matters for those who want to know the volume of a pleural effusion.
2. THE SIGNS OF PLEURAL EFFUSION
Traditionally, the diagnosis is based on an anechoic image. CEURF does not use this criterion in the critically ill. Only anechoic effusions are anechoic. How about the others, which can have all degrees of echogenicity, especially the most life-threatening: hemothorax, pyothorax, etc.? In addition, hard conditions (challenging patients) create parasite echoes with difficulties to affirm the anechoic pattern of the effusion. The CEURF definition has been made independent from the tone of the effusion. We first see a structural image (i.e., not an artifact) at the PLAPS- point. Structural images in the thorax, in critically ill patients, are of either pleural effusions or lung consolidations. What else? For defining the pleural effusion anyway and regardless of its volume, we use two signs of our own.
3. ONE STATIC SIGN: THE QUAD SIGN
This is the only static sign we use. A pleural effusion is limited by four regular borders shaping a quad. These borders are the pleural line, from where it arises; the upper and lower shadows of the ribs, regular as any artifact; and the deep border, which is always regular and roughly parallel to the pleural line (15° more or less), as it represents the lung surface. We imagine that apart from irregular pleural tumors that we never yet see, the lung surface is always regular. This line was called the lung line, an ultrasound marker of the visceral pleura. The lung line is visible when the visceral pleura is separated from the parietal pleura by a structure that allows ultrasound transmission, i.e., a fluid effusion. In healthy subjects, the lung line is virtual, making the parietal and the visceral pleura one line (the pleural line).
From the lung line, only the lung must be visible. It can appear as normal, yielding horizontal artifacts. It can yield vertical artifacts, called the sub-B-lines. It can yield lung consolidation (Fig. 4). If a heart happens to be seen in the depth, then only the question of a pseudo- pleural effusion and a real pericardial effusion may be raised. In 26 years, we never saw an effusion coming up to the lower extension of the PLAPS-point with a sharp angle and belonging to a pericardial sac.

Fig. 4 Septated pleural effusion. Left PLAPS- point. The lung line ( plain arrows, right arrow at a distance) demonstrates the pleural effusion. Septations are visible inside, indicating an infectious process. Deeper to the lung line, the lower lobe ( LL ) is consolidated. The cupola ( dotted arrows ) is completely motionless. The spleen ( S ) is far enough from the puncture site. Usual PLAPS: pleural and alveolar disorders in one same view.
Note: An aerated lung floats over the effusion. A consolidated lung floats within it (same density). The vision of the inferior part of the lung freely dancing within the effusion is reminder of alga, was coined the jellyfish sign, also “sirena tail” (suggested by Anne-Charlotte, from Tahiti 2005). Agnes Gepner gave a label which we could not assume. The jellyfish sign is just a variant of the sinusoid sign; see below (Fig. 5).
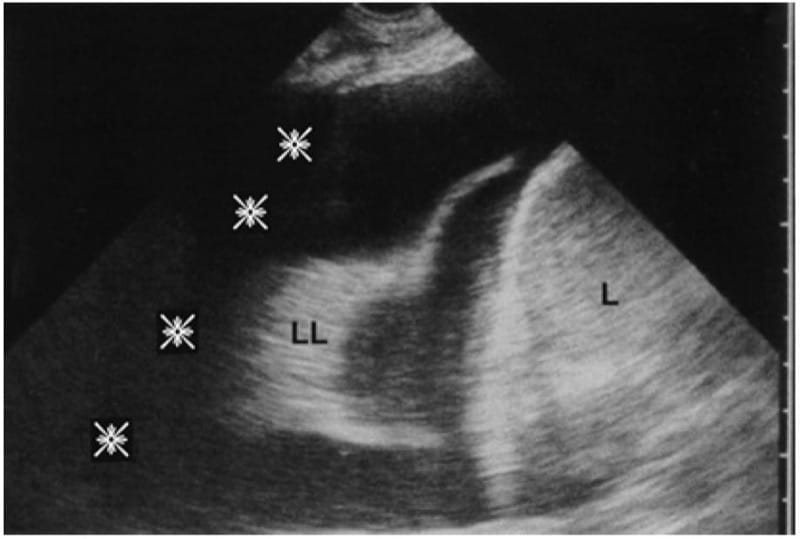
Fig. 5 Substantial pleural effusion. Intercostal route, longitudinal scan, PLAPS-point. The anechoic pattern just evokes the transudate but does not prove it. The lower lobe ( LL ) swims within the effusion in real time (yielding sinusoid sign). The BLUE-pleural index should be measured roughly at the lung line (i.e., here, 35 mm, indicating roughly 1,250–2,500 cc, slightly more if the lung consolidation is considered as having an index of 4, i.e., a correc-tion factor of 1.2, i.e., 1,500–3,000 cc). No measurement should be done below the lung, since it should be meaningless, going up to the mediastinum with a fixed distance (here more than 9 cm). L liver. Slight trick, the pleural effusion and the shadow of the rib ( asterisks ) are both anechoic.
Note: Sophisticated minds may ask how to distinguish a lung line from an A-line. First, the A-line is at a precise distance (the skin-pleural line distance). The A-line is strictly parallel to the pleural line. Just on a static image, a patient with a pleural effusion which would be located exactly at the same distance, and would be rigorously parallel to the pleural line, would be unlucky. In addition, the A-line is perfectly standstill, whereas the lung line usually has a dynamic: the sinusoid sign; see just below.
4. ONE DYNAMIC SIGN: THE SINUSOID SIGN
A gas can modify its volume under pressure, not a fluid. This is a basic rule in medicine, used when managing a cardiac arrest (from a talk with Boussignac, as we understood him). A pleural effusion in a rigid thorax, surrounding an aerated organ which inflates, follows this rule. This generates a dynamic sign, the respiratory variation of the interpleural distance. On inspiration, this distance decreases: the lung line moves toward the pleural line (Fig. 6). This sign indicates the inspiratory increase of size of the lung, spreading the fluid collection. As the lung moves toward a “core-surface” axis, the pattern, on M-mode, is a sinusoid. This sign, also quite specific to pleural effusion, is therefore slightly redundant with the quad sign. It is mainly relevant in two cases:
- In difficult examinations, when the quad sign is not easy to prove.
- Mainly, the sinusoid sign indicates a low viscosity. In very viscous or septate effusion, the sinusoid sign is absent. Minute effusions and the “butterfly syndrome”
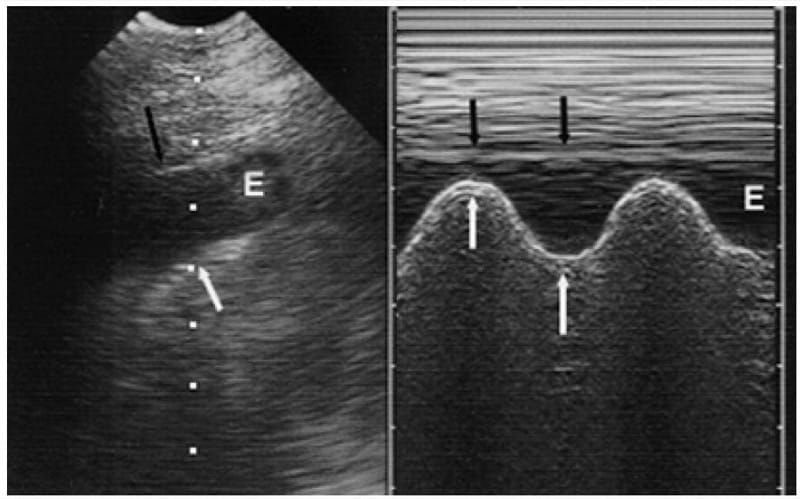
Fig. 6 The sinusoid sign. Left (real time). At the PLAPS-point, this collection’s thickness ( E ) varies in rhythm with the respiratory cycle. The lung line, deeper border (white arrows) moves toward the motionless pleural line ( black arrow ) shaping a sinusoid. The sinusoid sign is specific to pleural effusion. Right (M-mode). This image shows the relative dynamic of the lung line ( white arrows ) and pleural line ( black arrows )
Other Signs?
We heard on the spinal sign (showing the spine when there is an ultrasound window), a sign we don’t use. We heard on signs allowing distinction between pericardial and pleural effusions (considering the location of the aorta), but don’t feel the need when using our described technique. We do not conceive how a pleural effusion could be confused with something else.
5. VALUE OF ULTRASOUND: THE DATA
The quad and sinusoid signs confirm the pleural effusion with a specificity of 97 % when the gold standard used is withdrawal of pleural fluid.
Sensitivity and specificity are both 93 % with CT as gold standard. Note that extremely small effusions generate the quad and sinusoid sign, those which can be missed on CT. This partly explains why our data are lower than 100 %, also raising the question of the pertinence of this gold standard.
6. DIAGNOSING MIXT CONDITIONS (FLUID AND CONSOLIDATION) AND DIAGNOSING THE NATURE OR THE VOLUME OF A PLEURAL EFFUSION: INTERVENTIONAL ULTRASOUND (THORACENTESIS)
Complicated patterns can be seen, and we imagine that novices may find difficulties in distinguishing echoic fluids from anechoic, necrotizing lung consolidations. Here one can open to expert approaches, but the principle of the BLUE- protocol is first, not to pay attention to this distinction, i.e., call it a PLAPS. Foremost, the puncture will tell which antibiotic should be given, either the needle comes in the pleural cavity or within a very consolidated lung. Using this philosophy gives to the present chapter a reasonable thickness ( developed details such as the ways to know the nature of a pleural effusion, including direct thoracentesis).
Regarding the assessment of the volume, for making this chapter short. Just note at this step that the slightest pleural effusion is taken into consideration, defining a positive PLAPS.
The BLUE-Protocol and the Diagnosis of Pneumonia explains why a pleural effusion has the meaning of a pneumonia in the sequence of the BLUE-protocol.
7. PSEUDO-PITFALLS
We don’t know any real pitfall.
An image appearing through the diaphragm during an abdominal approach can be due to pleural fluid but also compact alveolar consolidation or the ghost of subphrenic organs (spleen, liver). Like all concave structures, the diaphragm can reflect (reverberate) underlying structures at the upper location (generating genuine ghosts) (Fig. 7). The solution for avoiding this pitfall is to forget this abdominal technique.
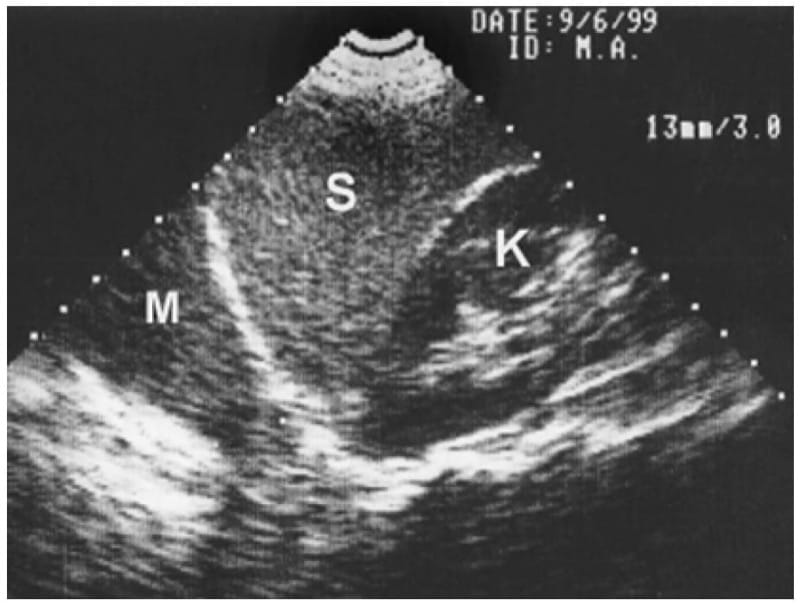
Fig. 7 A ghost. On this longitudinal subcostal scan, the left kidney ( K ), the spleen ( S ), the hemidiaphragm, and then an area ( M ) evoking pleural effusion can be observed. This mass M has a structure a bit too close to the spleen. This can be a ghost generated by the spleen refl ected by the diaphragm, a concave structure. Direct intercostal scans make these ghosts vanish.
By carefully detecting the quad sign, the user avoids to perpetrate a major error: diagnosing “pleural effusion” (meaning, by the way, inserting a needle in it) when fluid is seen. A stomach full of fluid and touching the wall, below the cupola (not far from the PLAPS-point), or, much worse, an ectopic stomach within the thorax create “fluid” collections. These collections, as a rule heterogeneous, may evoke the empyema. Here, there is no lung line. The deep boundary is scalloped (the gastric wall). In addition, when there is an air-fluid level, a typical “swirl sign” can be generated (see Fig. 7). The swirl sign shows the freedom of this air-fluid collection at atmospheric pressure, whereas a pleural effusion is the prisoner of the pleural pressure (apart from pneumothorax, etc.).
A picturesque pseudo-pitfall is the anechoic collection of silicone that we can find within certain breasts. Read Fig. 8 caption for knowing, if needed, the tricks for not falling under the charm of this troubling confusion (Fig. 8).
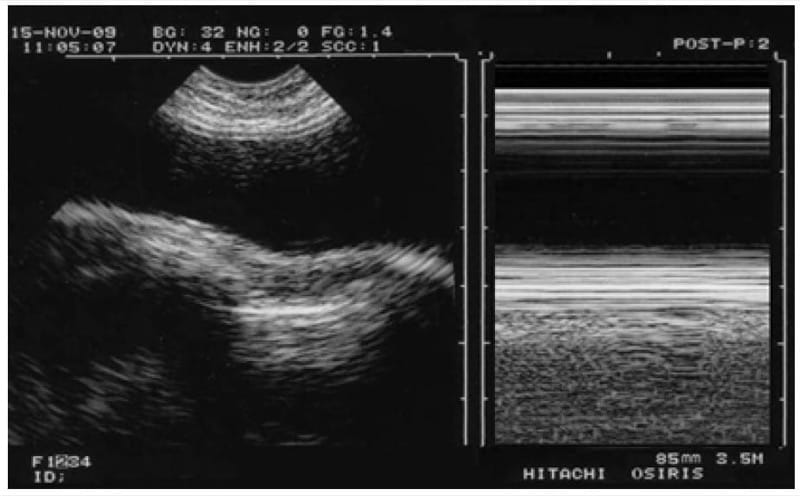
Fig. 8 An odd pleural effusion? A charming confusion. Hasty users, when diagnosing here a pleural effusion, would violate at least two principles of lung ultrasound. Principle N°2: a pleural effusion would not be sought for anteriorly (apart from rarities). Principle N°4: always begin by the bat sign, for not being abused by this silicone breast here. The pleural line is clearly visible below the “effusion,” with in addition a marked lung sliding at the right on M-mode (seashore sign).
8. ADDITIONAL NOTES ON PLEURAL EFFUSIONS
Abundant effusions allow analysis of deep structures (lung, mediastinum, descending aorta). One must take advantage of this effusion to explore them before evacuation: a ruptured descending aortic aneurism can be detected.
Does a pleural effusion abolish lung sliding? Of course it does, even a millimetric effusion.
Pleural Effusion: Some Main Points
Our 5-MHz microconvex probe is perfect in the adult (12 MHz in the newborn). One should forget the subcostal route. Search for small effusions first at the PLAPS-point. The main sign: regular deep limit (the quad sign). Slightly more accessory sign: the lung line moves toward the pleural line on inspiration (sinusoid sign). The echo-genicity is usually dark (anechoic), but echoic effusions are straightforward detected using these universal signs.