INTRODUCTION
Pectoralis nerve (Pecs) and serratus plane nerve blocks are newer ultrasound (US)-guided regional anesthesia techniques of the thorax. The increasing use of ultrasonography to identify tissue layers and, particularly, fascial layers has led to the development of several newer interfascial injection techniques for analgesia of the chest and abdominal wall. For instance, the Pecs I nerve block was devised to anesthetize the medial and lateral pectoral nerves, which innervate the pectoralis muscles.
This is accomplished by an injection of local anesthetic in the fascial plane between the pectoralis major and minor muscles. The Pecs II nerve block (which also includes the Pecs I nerve block) is an extension that involves a second injection lateral to the Pecs I injection point in the plane between the pectoralis minor and serratus anterior muscles with the intention of providing block of the upper intercostal nerves. A further modification is the serratus plane nerve block, in which local anesthetic is injected between the serratus anterior and latissimus dorsi muscles.
These interfascial injections were developed as alternatives to thoracic epidural, paravertebral, intercostal, and intrapleural nerve blocks, primarily for analgesia after surgery on the hemithorax. Initially, Pecs nerve blocks were intended for analgesia after breast surgery; however, case reports have also described the use of Pecs and serratus plane nerve blocks for analgesia following thoracotomy and rib fracture. Information from the currently published literature on Pecs and serratus plane nerve blocks in peer-reviewed journals is summarized in Table 1.
TABLE 1. Summary of published controlled clinical trials and case reports.
| Author, Year | Study Type | Surgery/Indication | Block Type | N | Injectate | Outcome |
|---|---|---|---|---|---|---|
| Blanco et al., 2013 | Volunteer study | – | Serratus plane | 4 | 0.4 mL/kg levobupivacaine 0.125% and gadolinium | Mean duration of paresthesia in the intercostal nerve distribution T2–T9, was 752 minutes (injection superficial to serratus anterior) |
| Wahba and Kamal, 2014 | Randomized controlled trial | Mastectomy | Pecs II versus PVB | 60 | 0.25% levobupivacaine: 15–20 mL T4 PVB, 10 mL Pecs I block | Pecs blocks reduced postoperative morphine consumption (first 24 h) and pain scores (first 12 h) in comparison with PVB following mastectomy |
| Fujiwara et al., 2014 | Case report | Insertion of cardiac resynchronization device | Intercostal at first and second interspace, Pecs I block | 1 | 0.375% ropivacaine: 4 mL intercostal block, 10 mL Pecs I block | Surgery performed under intercostal/ Pecs I blocks and dexmedetomidine |
| Kunhabdulla et al., 2014 | Case report | Analgesia for rib fracture | Serratus plane | 1 | 20 mL bolus 0.125% bupivacaine, then infusion of 0.0625% bupivacaine at 7–12 mL/h | Effective analgesia to enable physiotherapy and ambulation |
| Madabushi et al., 2015 | Case report | Analgesia for thoracotomy | Serratus plane | 1 | 6 mL bolus 1% lignocaine, then infusion of bupivacaine 0.1% at 7 mL/h | Improvement in pain and ventilation |
| Murata et al., 2015 | Case report | Breast surgery | Pecs II | 2 | 35 mL 0.2% ropivacaine (mastectomy); 45 mL 0.2% ropivacaine (lumpectomy) | Mastectomy performed under Pecs II block and supplemental infiltration |
| Ueshima, 2015 | Case report | Segmental breast resection | TTP combined with Pecs II | 1 | 0.15% levobupivacaine: 15 mL TTP, 10 mL Pecs I, 20 mL Pecs II | Surgery performed under TTP and Pecs II blocks |
| Bashandy and Abbas, 2015 | Randomized controlled trial | Mastectomy | Pecs II | 120 | 0.25% bupivacaine: 10 mL Pecs I, 20 mL Pecs II | Lower visual analog scale pain scores and opioid requirements in the Pecs group compared to control group |
| Kulhari, 2016 | Randomized controlled trial | Radical mastectomy | Pecs II versus PVB | 40 | 25 mL 0.5 % ropivacaine | Duration of analgesia increased in Pec’s block compared to PVB group (4.9 versus 3.3 hours) |
| Hetta, 2016 | Randomized controlled trial | Radical mastectomy | Serratus plane | 64 | 30 mL 0.25 % bupivacaine, Serratus plane; 15 mL 0.25% bupivacaine, PVB | Increased opioid consumption in the serratus plane compared to the PVB group |
Pecs nerve blocks have also been proposed in letters to the editor as alternative techniques to anesthetize operative regions such as the axilla, proximal medial upper arm, and posterior shoulder, which are not innervated by the brachial plexus (Figure 1).
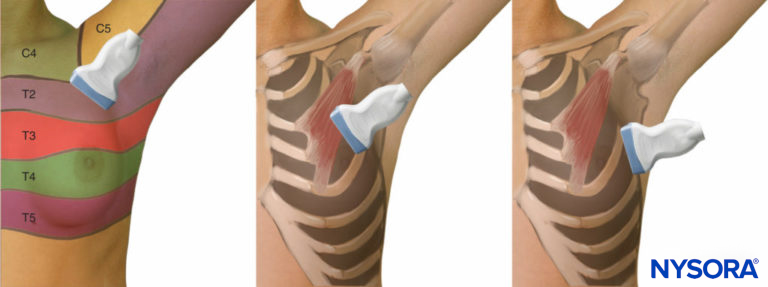
Figure 1. Transducer position for Pecs blocks. Also, refer to Figures 9 and 11.
ANATOMY OF THE PECTORAL AND AXILLARY REGIONS
Pecs nerve blocks are applied in the pectoral and axillary regions, with the muscles in both regions innervated by the brachial plexus. The pectoral region overlies the pectoralis major muscle and is limited by the axillary, mammary, and inframammary regions (Figure 2).
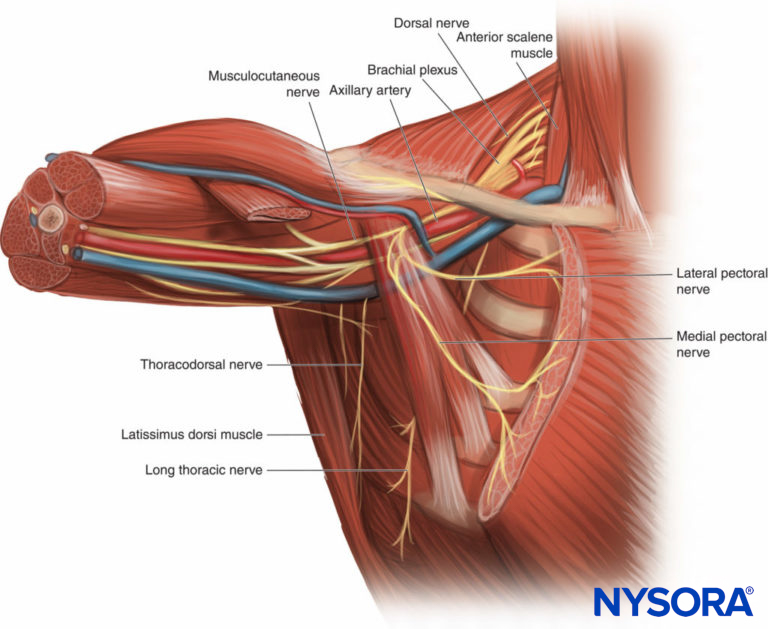
Figure 2. Pectoral region.
The axillary region is lateral to the pectoral region and consists of the area of the upper chest that surrounds the axilla. In both regions, there are muscles, nerves, and vessels within the fascial layers (Figure 3). In the pectoral region, there are four muscles relevant to Pecs nerve blocks: the pectoralis major, pectoralis minor, serratus anterior, and subclavius muscles. The pectoralis major and minor muscles are innervated by the lateral and medial pectoral nerves; the serratus anterior is innervated by the long thoracic nerve (C5, C6, and C7); and the subclavius is innervated by the upper trunk of the brachial plexus (C5 and C6).
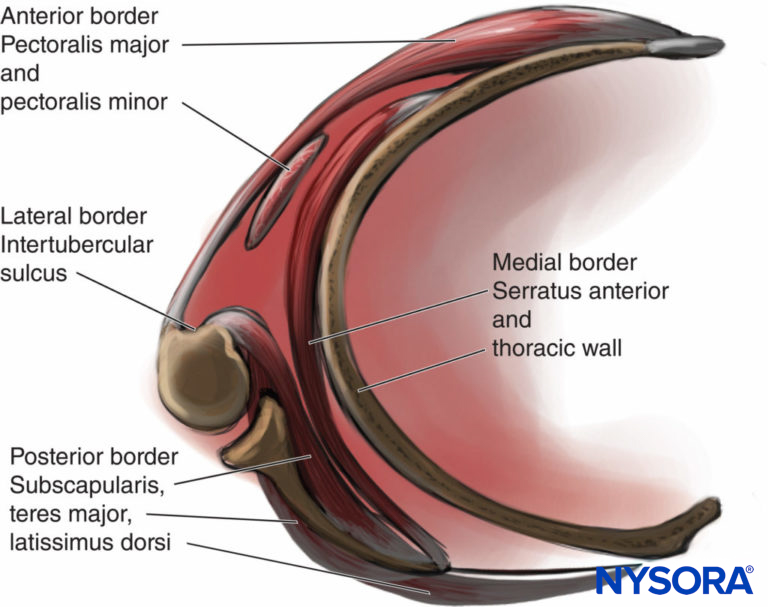
Figure 3. Axillary region.
The axillary region is a pyramidal structure with four borders:
1. The apex or axillary inlet, formed by a lateral border of the first rib, superior border of the scapula, and the posterior border of the clavicle
2. The anterior border, formed by the pectoralis major and minor muscles
3. The lateral border, formed by the humerus
4. The posterior border, formed by the teres major, latissimus dorsi, and subscapularis muscles.
Figure 2, the muscles, nerves, and vessels relevant to Pecs and serratus plane nerve blocks are summarized in Tables 2, 3 and 4 respectively.
TABLE 2. Nerves relevant to pecs and serratus plane nerve blocks.
| Nerve | Origin | Innervation | Relevance |
|---|---|---|---|
| Long thoracic | Roots (C5, C6, C7) | Serratus anterior muscle | Known as the nerve to the serratus anterior. |
| Lateral pectoral | Lateral cord (C5, C6, C7) | Pectoralis major and pectoralis minor muscles | Penetrates the clavipectoral fascia to supply the pectoralis major directly and, through communication with the medial pectoral nerve, the pectoralis minor. There is no cutaneous branch. Can be located on the deep surface of the pectoralis major. |
| Medial pectoral | Medial cord (C8, T1) | Pectoralis major and pectoralis minor muscles | Penetrates the deep surface of the pectoralis minor to innervate this muscle before penetrating it to supply the pectoralis major muscle. |
| Intercostal | Anterior rami of thoracic spinal nerves | Segmental somatic sensory innervation to skin | Lateral cutaneous branches of T2-T6 innervate the lateral breast. Accessible in the mid-axillary line. |
| Thoracodorsal | Posterior cord (C6, C7, C8) | Latissimus dorsi muscle | Large nerve from the posterior cord, which has a course in the posterior axillary wall, crosses the lower border of the teres major to enter the deep surface of the latissimus dorsi muscle. The thoracodorsal nerve is adjacent to the thoracodorsal artery. |
TABLE 3. Muscles relevant to pecs and serratus plane nerve blocks.
| Muscle | Innervation | Relevance |
|---|---|---|
| Pectoralis major | Medial (C8, T1) and lateral (C5–C7) pectoral nerves | Sonographic landmark. |
| Pectoralis minor | Both pectoral nerves (C5–C8) | Sonographic landmark. |
| Serratus anterior | Long thoracic nerve (C5–C7) | Sonographic landmark for Pecs II and serratus plane blocks. The intercostobrachial, long thoracic, and thoracodorsal nerves lie on this muscle. The thoracodorsal artery is superficial to this muscle. |
| Teres major | Subscapular nerve (C5–C6) (offspring of subscapularis muscle) | Contributes to the posterior wall of the axilla. |
| Subscapularis | Upper and lower subscapular nerves (C5–C8) | Contributes to the posterior wall of the axilla. |
| Latissimus dorsi | Thoracodorsal nerve (C6–C8) | Contributes to the posterior wall of the axilla; sonographic landmark for serratus plane blocks. |
TABLE 4. Vessels relevant to pecs and serratus plane nerve blocks.
| Vessel | Relevance |
|---|---|
| Axillary | Is the continuation of the subclavian artery after it passes the lateral border of the first rib. It lies lateral to the axillary vein. It gives of the branches listed below. |
| Superior thoracic | Branch of the first part of the axillary artery; supplies both pectoral muscles. |
| Thoracoacromial | Arises from the second part (deep to the pectoralis minor) of the axillary artery, close to the upper border of the pectoralis minor; pierces the clavipectoral fascia in the infraclavicular fossa; has four branches that may arise deep or superficial to the clavipectoral fascia. |
| Lateral thoracic | Follows the lower border of the pectoralis minor; supplies both pectoral muscles. |
| Thoracodorsal | Arises from the third part (distal to the pectoralis minor) of the axillary artery; initially known as the subscapular artery, located in the posterior axillary wall (largest branch of the axillary artery), before becoming the thoracodorsal artery; has a course with the thoracodorsal nerve, which innervates the latissimus dorsi. |
The pectoral and axillary regions are separated by fascias. In the pectoral region, there are two main fascias: the superficial fascia and the deep thoracic fascia. The deep thoracic fascia divides into three separate fascias: the pectoral (superficial), clavipectoral (intermediate), and exothoracic (deep). The clavipectoral fascia stretches between the clavicle and the pectoralis minor (Figure 4) and encloses the pectoralis minor with a thin layer of fascia. Between the pectoralis minor and subclavius muscles, the two layers of the clavipectoral fascia fuse.
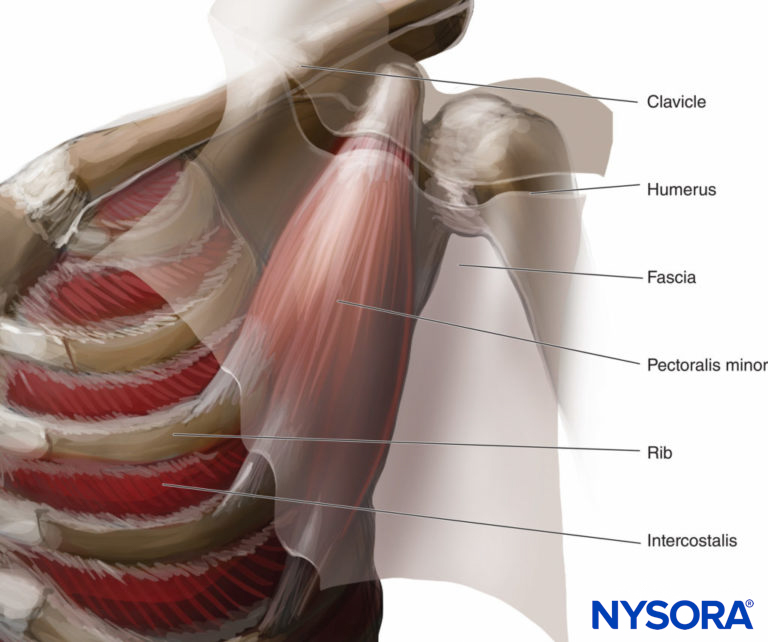
Figure 4. The clavipectoral fascia and its continuation into the axillary fascia.
Caudal to the pectoralis minor, the clavipectoral fascial layers rejoin to form the suspensory ligament of the axilla, which is joined to the axillary fascia (Figure 5).
At the pectoral level, the fascias create four potential compartments for the injection of local anesthetic:
- Between the superficial and deep pectoral fascial layers
- Between the pectoral fascia and the clavipectoral fascia
- Between the clavipectoral fascia and the superficial border of the serratus anterior muscle
- Between the serratus anterior muscle and the exothoracic fascia
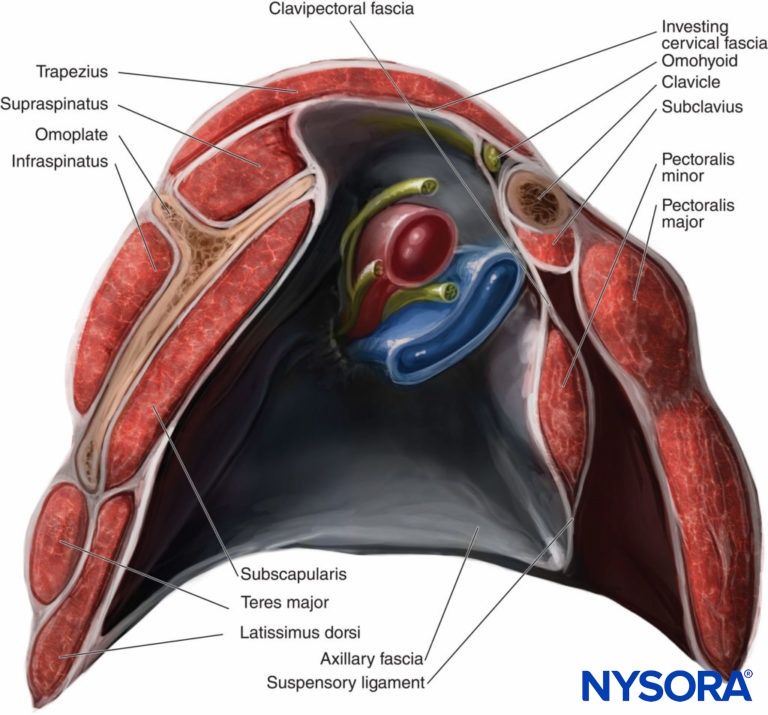
Figure 5. Section of the axilla showing the clavipectoral fascia enclosing the subclavius and the pectoralis minor muscles. Inferior to the pectoralis minor muscle, the clavipectoral fascia becomes the suspensory ligament.
The first two compartments are in the pectoral region, but the third and fourth communicate with the axillary region. The nerves and vessels in this region create communications by crossing the compartments. The nerves of the pectoral region are mainly the lateral and medial pectoral nerves, but there is also an important innervation from the supraclavicular nerve and from the lateral and anterior branches of the intercostal nerves. The lateral pectoral nerve crosses the axillary artery anteriorly and pierces the clavipectoral fascia in close relationship with the thoracoacromial artery on the undersurface of the upper portion of the pectoralis major muscle, which it supplies with lateral cord fibers from C5–C7 (Figure 6).
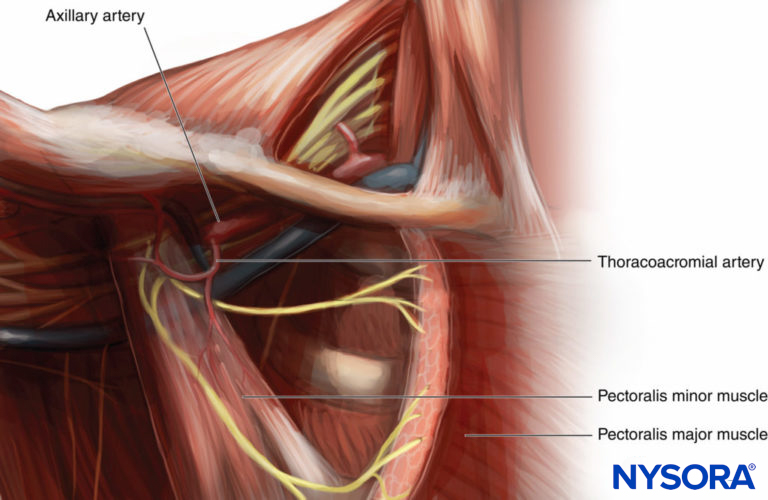
Figure 6. Relationship of the lateral pectoral nerve to the acromiothoracic artery.
The lateral pectoral nerve is medial to the pectoralis minor before entering the pectoralis major muscle; it communicates across the axillary artery with the medial pectoral nerve and, through this communication (via ansa pectoralis), supplies the pectoralis minor. The medial pectoral nerve arises from the medial cord fibers from C8–T1, behind the axillary artery at the level below the clavicle, and passes through the deep surface of the pectoralis minor, which is perforated and then enters and innervates pectoralis major. Both pectoral nerves enter the deep surface of the pectoralis major, and neither has a cutaneous branch. The nerves of the axillary region are the intercostobrachial, intercostal T3–T9, long thoracic, and thoracodorsal. The intercostobrachial nerve is the lateral cutaneous branch of the second and third intercostal nerves in 67% and 33% of cases, respectively. It crosses the serratus anterior muscle in the midaxillary line to innervate the axilla. The intercostobrachial nerve is a vital nerve if regional anesthesia of the axilla is required.
The intercostal nerves (T3–T9) provide motor supply to the intercostal muscles and receive sensory information from the skin and parietal pleura. The intercostal nerves have posterior, lateral, and anterior branches and an anterior accessory branch that innervates the sternum. The lateral branches innervate most of the pectoral and axillary regions, together with the posterior hemithorax, back to the scapula. They pierce the external intercostal muscle and exit between the serratus anterior digitations at the level of the midaxillary line. The long thoracic nerve is in the axillary compartment close to the lateral thoracic branch of the thoracoacromial artery and travels down the lateral aspect of the serratus anterior muscle, which it innervates.
Arising from the posterior cord, the thoracodorsal nerve, C6–C8 (nerve to the latissimus dorsi), has a course posteriorly in the axillary compartment, in close relationship with the thoracodorsal artery (Tables 2 through 4). The thoracodorsal nerve becomes prominent when the humerus is abducted and laterally rotated. It is an important and large nerve in danger during reconstructive surgery and other operations involving the lower axilla (see Table 2).
For a more comprehensive review, see Functional Regional Anesthesia Anatomy.
THORACIC WALL NERVE BLOCKS
Pecs I Nerve Block
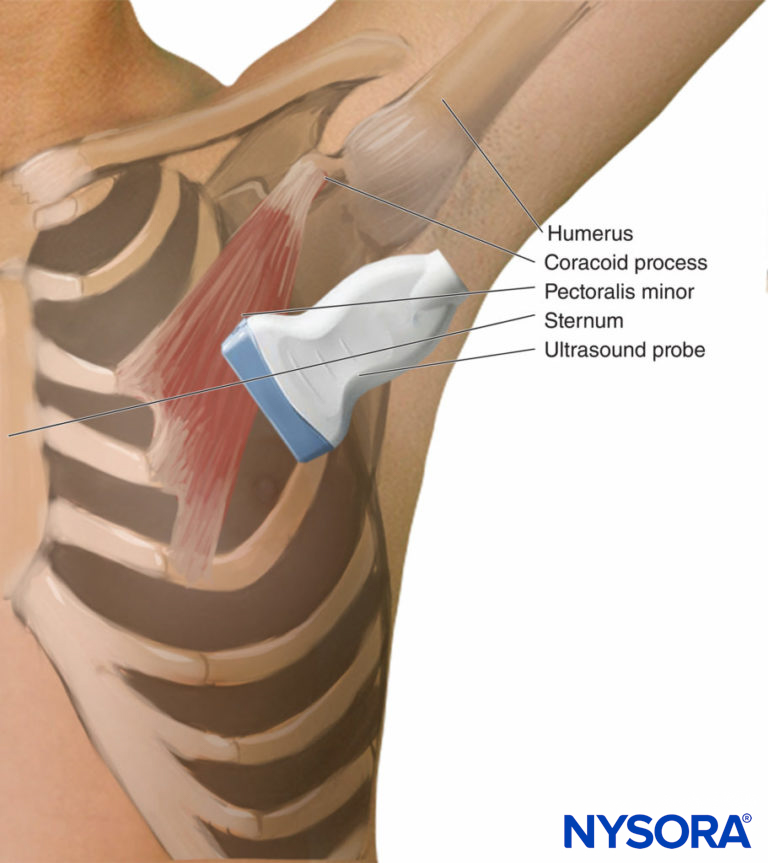
The Pecs I nerve block involves a hydrodissection of the plane between the pectoral muscles with local anesthetic to nerve block the lateral and medial pectoral nerves. The main landmarks to identify the point of injection under US guidance are the pectoralis major and pectoralis minor muscles and the pectoral branch of the thoracoacromial artery. The nerve block is performed with the patient supine, either with the arm next to the chest or abducted 90 degrees. With standard American Society of Anesthesiology (ASA) monitoring and supplemental oxygen, the operator locates the coracoid process on US in the paramedian sagittal plane. The transducer is rotated slightly to allow an in-plane needle trajectory from the proximal and medial side toward the lateral side (ie, the caudal border of the transducer is moved laterally, while the proximal border remains unchanged) (Figure 7). This rotation helps image the pectoral branch of the thoracoacromial artery. The proper fascial plane is confirmed by hydrodissection to open the space between the pectoralis muscles. The suggested volume is 0.2 mL/kg of a long-acting local anesthetic (Figure 8 however, the reader should be informed that dose-ranging studies have not been conducted at the time of publication, and, therefore, there are no evidence-based recommendations currently available.

From the Regional Anesthesia Manual: Reverse Ultrasound Anatomy for a PEC I block with needle insertion in-plane and local anesthetic spread (blue). PTA, pectoral branch of the thoracoacromial artery; R2, second rib; R3, third rib.
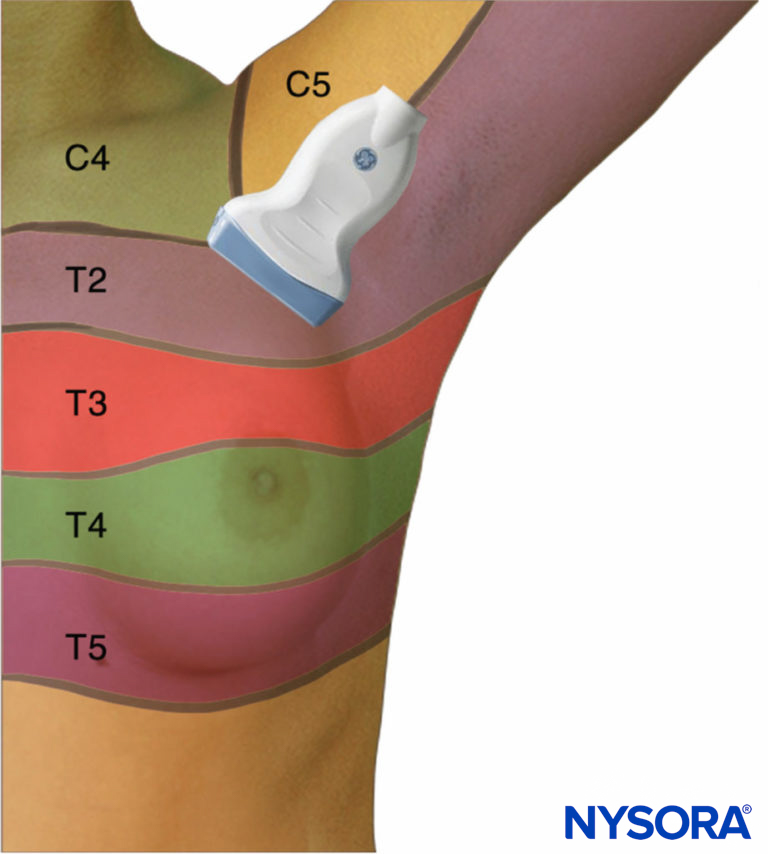
Figure 7. Transducer position for the Pecs I nerve block.
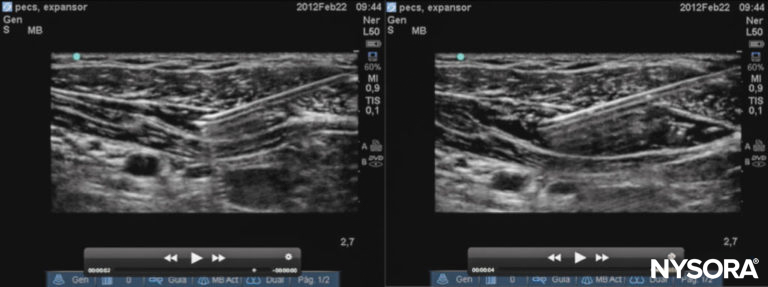
Figure 8. Sonogram of the Pecs I injection. Left: needle placement; right: desired spread of local anesthetic.
Pecs II Nerve Block
The goal of the Pecs II nerve block is to infiltrate two fascial compartments by dividing the dose of local anesthetic between the pectoral nerves (the pectoral fascia and clavipectoral fascia) and under the pectoralis minor muscle (between the clavipectoral fascia and the superficial border of the serratus muscle). The local anesthetic should cover two important compartments of the fascias involved: The pectoral compartment with the pectoral nerves and the intercostal branches for the axilla and chest. The nerve block is performed with the patient supine, either with the arm abducted 90 degrees or by his or her side. The first injection is similar to Pecs I, whereas the second is made at the anterior axillary line at the level of the fourth rib. The depth is usually 1–3 cm for the first injection and 3–6 cm for the second injection. With the transducer at the midclavicular level and angled infero-laterally, the axillary artery and vein and the second rib can be identified (Figure 9).
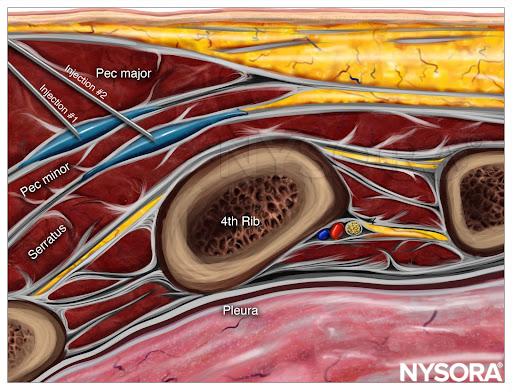
From the Regional Anesthesia Manual: Reverse Ultrasound Anatomy for a PEC II block with needle insertion in-plane and local anesthetic spread (blue) 1) Between the pectoralis major and minor muscles (PEC I), and 2) Between the pectoralis minor and serratus anterior muscles.
The transducer is then moved laterally until the pectoralis minor and serratus anterior are identified. With further lateral transducer movement, the third and fourth rib can then be identified. The local anesthetic is injected at two points: The first injection of approximately 0.2 mL/kg long-acting local anesthetic is made between the pectoral major and minor muscles, and the second injection of 0.2 mL/kg is made between the pectoralis minor and serratus anterior muscles. Figure 10 illustrates the sonographic anatomy, needle trajectory, and the desired spread of injectate.
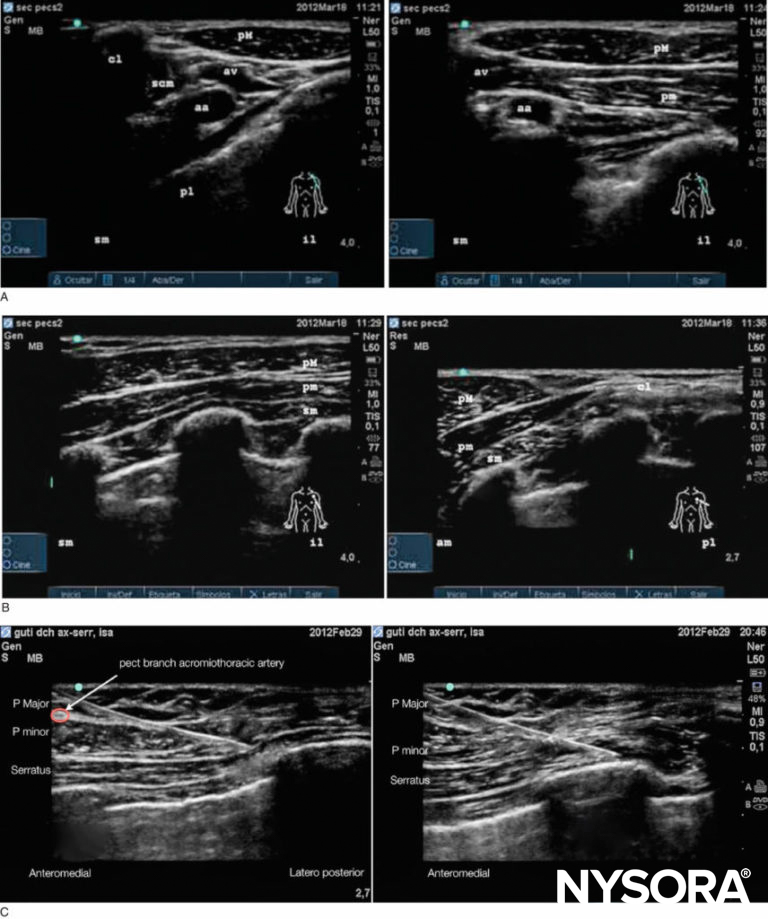
Figure 10. Pecs II sonogram: steps to locate points of injection. A: Left: Start from the clavicle; right: Count ribs down to the axilla. B: Left: First injection between the pectoralis major and pectoralis minor; right: Angle probe to locate Gilbert’s ligament. C: Left: Above the serratus muscle; right: Underneath the serratus muscle; cl, clavicle; scm, subclavius muscle; pM, pectoralis major; pm, pectoralis minor; av, axillary vein; aa, axillary artery; pl, pleura; sm, serratus muscle.
Serratus Anterior Plane Nerve Block
The serratus plane nerve block is performed in the axillary region, at a more lateral and posterior location than the Pecs I and II nerve blocks. At the axillary fossa, the intercostobrachial nerve, lateral cutaneous branches of the intercostal nerves (T3–T9), long thoracic nerve, and thoracodorsal nerve are located in a compartment between the serratus anterior and the latissimus dorsi muscles, between the posterior and midaxillary lines.
The two main anatomical landmarks are the latissimus dorsi and the serratus anterior muscles. The thoracodorsal artery runs in the fascial plane between the two. The ribs, pleura, and intercostal muscles can also be seen during the procedure. Lying on the side or supine with the arm brought forward is the preferable patient position. There are two main methods for identifying the plane for the serratus nerve block. The first method requires counting the ribs from the clavicle while moving the transducer laterally and distally until the fourth and fifth ribs are identified (Figure 10). The transducer is orientated in the coronal plane and then tilted posteriorly until the latissimus dorsi (a superficial thick muscle) is identified (Figure 11). The serratus muscle, a thick, hypoechoic muscle deep to the latissimus dorsi is imaged over the ribs. Translating the transducer posteriorly facilitates the identification of the plane between the serratus anterior and latissimus dorsi muscles. An alternative method is to place the transducer across the axilla, where the latissimus dorsi will appear more prominent (Figure 12). The location of the thoracodorsal artery is easier to identify this way. Both in-plane and out-of-plane approaches are appropriate. Following the identification of sonographic landmarks, regional anesthesia can be achieved using a 38-mm, 6–13-MHz, linear transducer set for small parts and a depth of 1–4 cm; a 50–100-mm, 22-gauge regional nerve block needle; and an injectate of 0.4 mL/kg of long-acting local anesthetic.
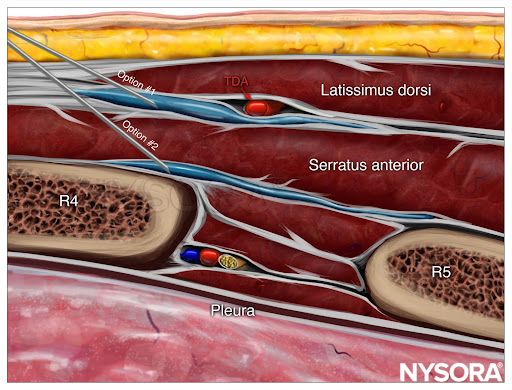
From the Regional Anesthesia Manual: Reverse Ultrasound Anatomy for a serratus anterior plane block with needle insertion in-plane and local anesthetic spread (blue) in option 1 (between the latissimus dorsi and serratus anterior muscles) or option 2 (underneath the serratus anterior muscle). TDA, thoracodorsal artery.
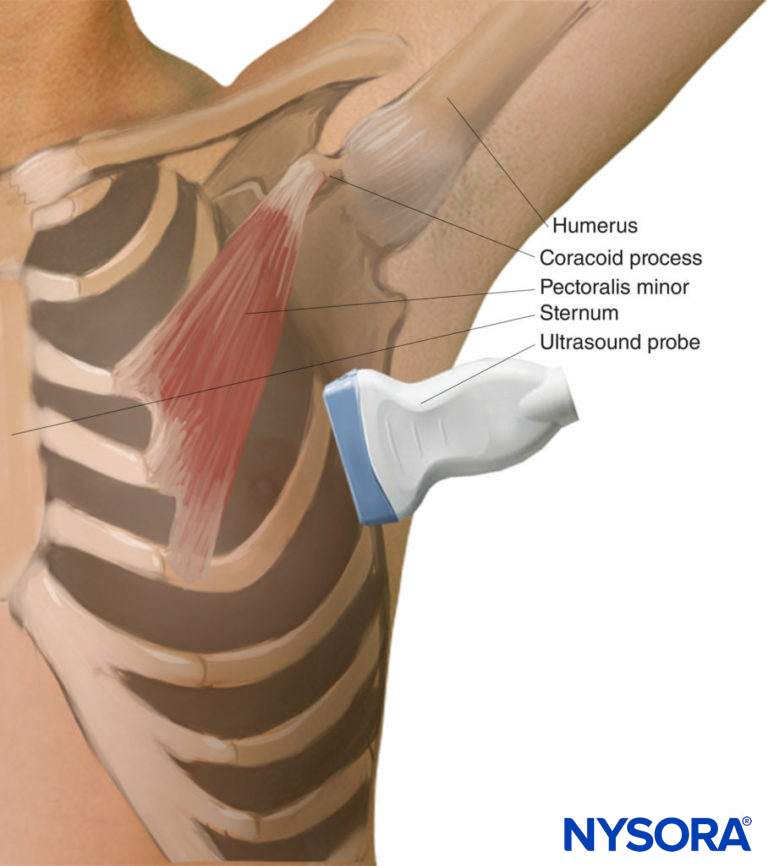
Figure 11. Transducer position required for the serratus plane nerve block.
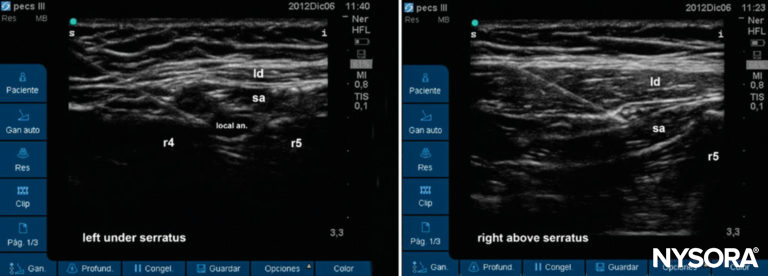
Figure 12. Sonogram of two possible levels for the serratus plane nerve block below (left) or above the muscle (right).
THE ANALGESIC POTENTIAL OF PECS NERVE BLOCKS
A summary of published studies is given in Table 1. At the time of writing, there were two randomized controlled trials (180 patients), five case reports (6 patients), and one volunteer study (4 patients, 8 hemithoraces). Bashandy and Abbas reported lower visual analog scale pain scores and opioid requirements in the Pecs group compared to a control group of patients following mastectomy. In this study, there was an inadequate description of allocation concealment and blinding of operating room personnel. Wahba and Kamal compared Pecs nerve blocks to paravertebral nerve blocks in 60 patients undergoing mastectomy. They found that Pecs nerve blocks reduced postoperative morphine consumption (in the first 24 hours) and pain scores (in the first 12 hours) in comparison with paravertebral block following mastectomy. Pecs nerve blocks have also been used for the insertion of a cardiac resynchronization device. The few remaining reports describe the utility of serratus plane nerve blocks for analgesia following rib fracture and thoracotomy.
SUMMARY
Pecs and serratus plane nerve blocks are newer US-guided nerve blocks for analgesia after breast and lateral thoracic wall surgery. The key sonographic landmarks are the pectoralis major, pectoralis minor, and serratus anterior muscles and the pectoral branch of the acromiothoracic artery. As there are currently few reports on the utility of these US-guided interventional analgesia modalities, data from the imminent randomized controlled trials will be necessary to establish the analgesic benefit, indications, and safety of the Pecs and serratus plane nerve blocks.
Clinical updates
Azem et al. (Regional Anesthesia & Pain Medicine, 2025) report in a retrospective exploratory study of lung transplantation that adding superficial parasternal intercostal plane (sPIP) and serratus anterior plane (SAP) blocks to standard care was associated with earlier tracheal extubation, with 16% of patients extubated within 8 hours compared with none in controls. The combined blocks also produced a significant reduction in opioid consumption, lowering 24-hour morphine equivalents and cumulative opioid use through 72 hours, without block-related complications. These findings suggest sPIP + SAP blocks are a safe and effective alternative when thoracic epidural analgesia is contraindicated, particularly in lung transplantation involving ECMO.
- Read more about the study HERE.
Bailey et al. (British Journal of Anaesthesia, 2025) report in a double-blind feasibility trial that continuous bilateral serratus anterior plane (SAP) catheters with ropivacaine do not improve pain scores, opioid consumption, or quality of recovery after adult cardiac surgery via median sternotomy compared with placebo. The study was deemed not feasible for progression at a single centre due to low recruitment and a high pneumothorax rate, with longer hospital stays among affected patients. These findings suggest that SAP catheters are unlikely to be effective or safe for routine sternotomy analgesia, and future research should focus on alternative parasternal or thoracic-plane techniques.
- Read more about the study HERE.
Jackson et al. (Anesthesiology, 2024) report in a single-center randomized, blinded, placebo-controlled trial of 92 patients undergoing minimally invasive thoracoscopic lung resection that adding a serratus anterior plane (SAP) block to an enhanced multimodal analgesia pathway did not significantly reduce 24-hour opioid consumption in the intention-to-treat analysis. A sensitivity as-treated analysis showed a modest but statistically significant reduction in 24-hour IV morphine equivalents, and SAP block was associated with lower composite pain scores with cough, but no improvements in quality of recovery, inspiratory volumes, nausea/vomiting, or length of stay. These findings suggest SAP block offers limited incremental benefit when intercostal blocks and robust multimodal analgesia are already in place, warranting further study to define which patients may derive meaningful opioid-sparing effects.
- Read more about the study HERE.
Vandenbrande et al. (Regional Anesthesia & Pain Medicine, 2024) report in a double-blind randomized controlled trial that a combined deep and superficial single-injection serratus anterior plane block (SAPB) significantly reduces opioid consumption after totally endoscopic aortic valve replacement, lowering 24-hour opioid use by ~40% compared with standard care. SAPB also produced lower pain scores at 4, 8, and 24 hours, though it did not improve extubation time, ICU stay, or hospital length of stay. These findings support SAPB as an effective opioid-sparing chest wall block within ERACS pathways for minimally invasive cardiac valve surgery.
- Read more about the study HERE.
Grape et al. (Journal of Clinical Anesthesia, 2020) report in a systematic review, meta-analysis, and trial sequential analysis of 16 randomized trials (1026 patients) that PECS and serratus plane blocks reduce early postoperative pain after breast surgery, lowering rest pain scores at 2 hours and decreasing IV morphine consumption by ~7 mg at 24 hours compared with no block. PECS blocks also reduced PONV, with benefits consistent across mastectomy and breast-conserving surgery ,and firm evidence confirmed by trial sequential analysis. Analgesic effects were more pronounced when axillary lymph node dissection was not performed, supporting PECS blocks as a first-line regional technique within multimodal analgesia for higher-pain breast procedures.
- Read more about the study HERE.
The review of Pirri et al. (Current Opinion in Anesthesiology, 2024) clarifies that the effectiveness of fascial plane blocks depends less on simple “spread between layers” and more on the microanatomy of fascia, including collagen fiber orientation, interfascial connective tissue density, and embedded neural and vascular structures. The fascia is a dynamic, innervated tissue, not an inert compartment, which helps account for the variable onset, spread, and duration observed with blocks such as ESP, TAP, and QLB. The review highlights that local anesthetic spread occurs through a combination of hydrodissection, diffusion along collagen networks, and uptake by nociceptive and sympathetic fibers within fascia, rather than predictable bathing of named nerves. Clinically, this framework supports ultrasound-guided precision, lower injection pressures, and tailored volumes, while reinforcing why some fascial plane blocks provide analgesia rather than dense anesthesia. Overall, fascial plane blocks are biologically active regional techniques that guide more realistic expectations and safer, more consistent application in perioperative pain management.
- Read more about the study HERE.