A newly published, manufacturer-independent study from Sweden (Gatzinsky et al., RAPM 2025) has brought critical insights into the durability and effectiveness of spinal cord stimulation (SCS) and dorsal root ganglion (DRG) stimulation therapies used in treating chronic neuropathic pain. The findings, drawn from real-world patient data spanning a decade, show that device failure and removal (explantation) is more common over time than previously recognized.
With nearly 4 out of 10 devices explanted by the 10-year mark, the study raises important clinical, technological, and patient selection concerns.
Highlights from the study
- Total patients studied: 400
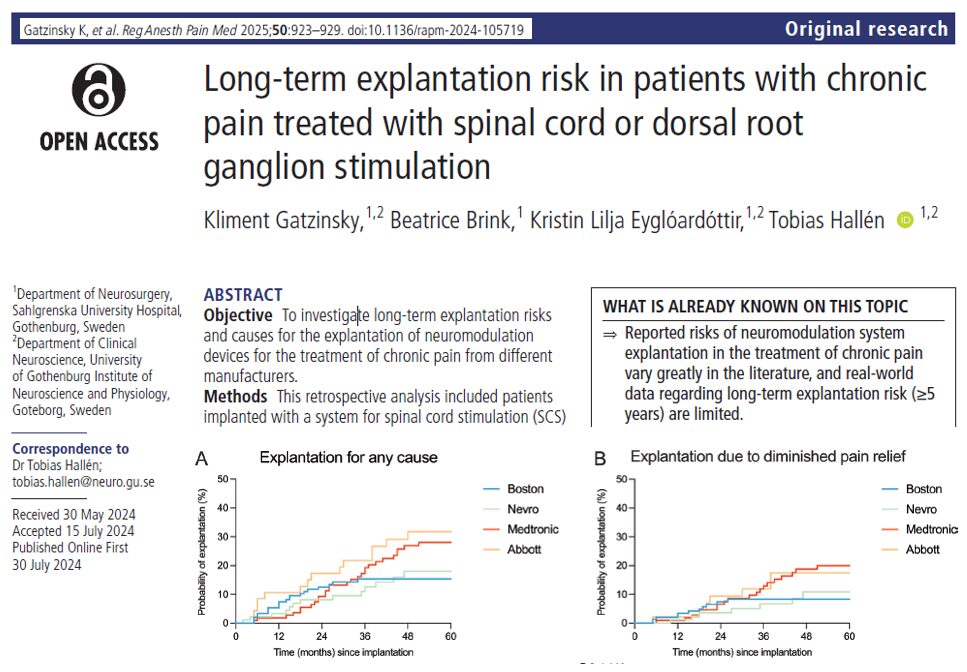
- Overall explantation rate: 24%
- 10-year cumulative explantation risk: 38%
- Primary cause of explantation: Loss of pain relief (55.2%)
- Reimplantation rate after removal: Only 25%
- DRG stimulation had the highest explantation risk over time
- Older Medtronic models are associated with higher failure
What is spinal cord stimulation?
SCS is a form of neuromodulation therapy where mild electrical impulses are delivered to the spinal cord to interfere with pain signals. The system includes:
- Electrodes implanted in the epidural space
- An implantable pulse generator (IPG)
- A remote control for patient programming
DRG stimulation is a more targeted variant where electrodes are placed over the dorsal root ganglion, a nerve structure that plays a key role in pain transmission, especially in localized (focal) neuropathic pain.
Benefits of neuromodulation:
- Reduces chronic, medication-resistant pain
- Improves quality of life
- Enables physical rehabilitation
- Can reduce opioid dependency
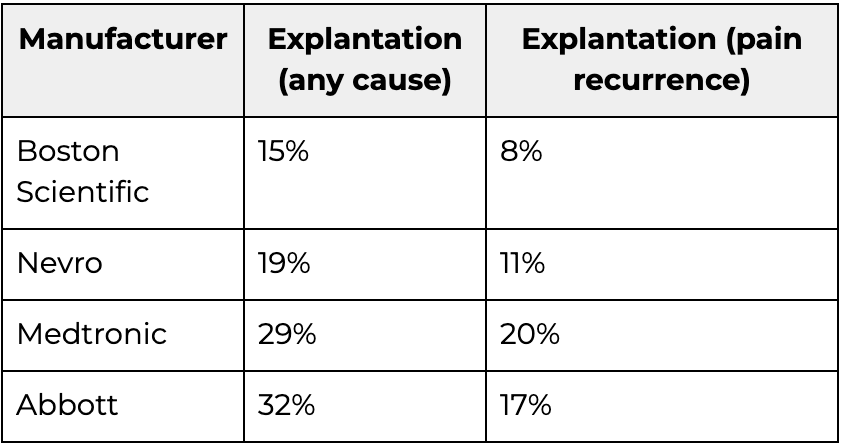
5-year explantation risks by manufacturer
DRG stimulation: high explantation risk
Among 16 patients implanted with DRG devices (all from Abbott):
- 5-year explantation risk: 38%
- 10-year explantation risk: 63%
- Main reason for removal: Diminished pain relief (63% of cases)
Multivariable regression analysis found DRG devices were 6.7 times more likely to be explanted due to pain recurrence (HR 6.73, p=0.03).
This suggests that while DRG may offer targeted therapy, it may also be prone to hardware challenges like lead breakage and dislocation, which are more common due to the anatomical location.
Why are devices removed?
The study classifies explantation causes as follows:
- Diminished pain relief: 55.2%
- MRI incompatibility: 17.7%
- Infection: 8.3%
- No longer needed: 4.2%
- Hardware issues (e.g., dislocation, pocket pain): 3.1%
- Other/unknown causes: 11.5%
MRI-related explantation was primarily due to non-MRI-conditional devices, particularly older Medtronic models. The trend is expected to decline with increased use of full-body MRI-safe implants.
How to reduce the risk of explantation
- Use modern devices that offer multiple waveforms and paresthesia-free options.
- Ensure MRI compatibility at the time of implantation to avoid removals later.
- Select patients carefully, particularly those with realistic expectations.
- Follow up closely to detect declining efficacy early and intervene.
- Refer to SCS-specific decision tools, such as the SCS e-health tool, for guidance on candidacy.
Clinical implications
- Device evolution matters: Newer models appear more durable and adaptable. Systems offering burst, high-frequency, or closed-loop stimulation may perform better over time.
- Manufacturer matters: While not statistically conclusive, devices from Boston Scientific and Nevro performed more favorably than older Medtronic and DRG systems from Abbott in this dataset.
- Real-world data is crucial: Unlike many manufacturer-funded studies, this independent review offers a less biased look at long-term therapy sustainability.
Subgroup analysis: PSPS2 patients
To reduce variability, the authors ran a subgroup analysis on the 241 patients with persistent spinal pain syndrome type 2 (PSPS2):
- Medtronic again showed a higher risk, but the difference wasn’t statistically significant (log-rank p=0.11 for any cause, p=0.15 for diminished pain relief).
Limitations of the study
The authors note several limitations:
- Retrospective design: Potential for missing data
- No consideration of revisions or partial failures, which are also clinically relevant
- Technological shifts during the study: Older Medtronic devices dominated early in the study; newer devices from Nevro and Boston Scientific came later
- Small DRG sample size (n=16): Makes broad conclusions difficult
- No evaluation of opioid use or psychological comorbidities, which could affect explantation decisions
Despite these constraints, the real-world data gathered provides valuable insight into long-term device performance and failure patterns.
Recommendations for future research
The authors call for:
- Large, prospective, multicenter trials using modern devices
- Independent registries free of manufacturer bias
- Studies on patient-reported outcomes, including quality of life, activity level, and satisfaction
- Technology-specific comparison trials (e.g., burst vs. 10kHz vs. DRG vs. traditional tonic stimulation)
Conclusion
While spinal cord and DRG stimulation remain vital tools in the chronic pain management arsenal, this study highlights a growing need to focus on long-term durability, device innovation, and smarter patient selection.
With nearly 1 in 4 devices removed within 5 years, and 1 in 3 within 10 years, the emphasis must shift from just successful implantation to ensuring sustained benefit and therapy longevity.
For more information, refer to the full article in RAPM.
Gatzinsky K, Brink B, Eyglóardóttir KL, Hallén T. Long-term explantation risk in patients with chronic pain treated with spinal cord or dorsal root ganglion stimulation. Reg Anesth Pain Med. 2025;50(12):923-929.
For more information on spinal cord stimulation and a pain medicine AI-assistant, download NYSORA’s Pain Medicine Assistant App!