Indications
- Preoperative assessment: In patients at risk or known to have phrenic nerve damage.
- Postoperative monitoring: After high-risk surgery, i.e., high abdominal or cardiothoracic surgery, or an interventional procedure that may result in unilateral diaphragm paresis (e.g., interscalene nerve blocks).
- Critical care setting, during ventilatory weaning: Assessment of work of breathing, diagnose atrophy of the diaphragm, and predict difficult ventilator weaning.
- Assessment of neuromuscular disorders
- Evaluation of respiratory diseases
Essential info
- Diaphragm ultrasound is non-invasive, highly accurate, and repeatable at the bedside.
- It can guide invasive procedures such as electromyography (EMG).
- It provides real-time information, allowing for dynamic assessment of the diaphragm’s movement during the respiratory cycle.
- Trained individuals have a sensitivity of 93% and specificity of 100% for diagnosing neuromuscular diaphragmatic dysfunction.
- Intra-observer correlation coefficients are as high as 87%-99%, and interobserver correlation may range from 56% to 98%.
Note
The choice between imaging modalities depends on the specific clinical context and the information needed for diagnosis and management.
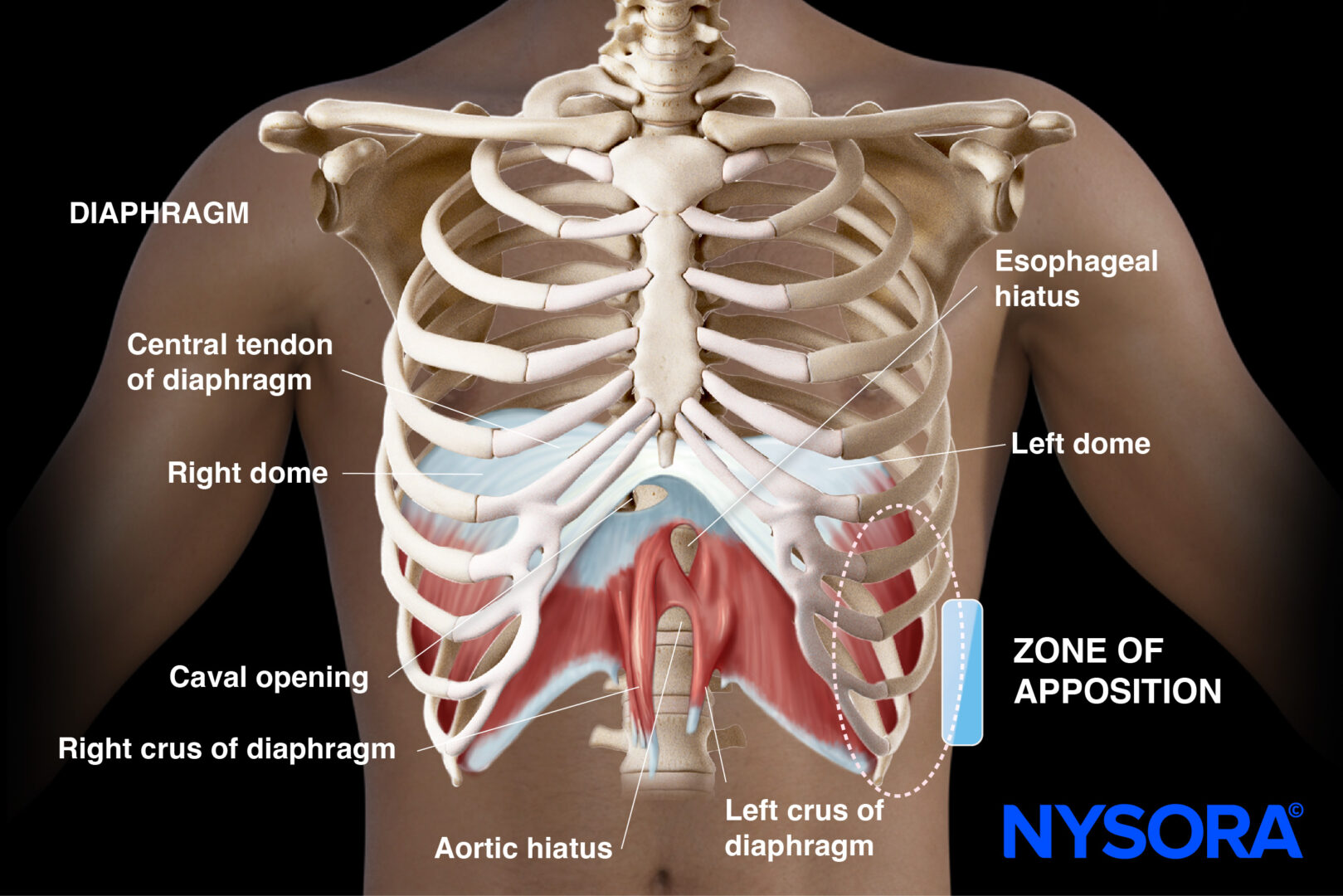
Functional anatomy
The diaphragm is a dome-shaped, musculotendinous structure separating the thoracic and abdominal cavities. It is the primary muscle involved in the process of breathing and plays a crucial role in the respiratory system.
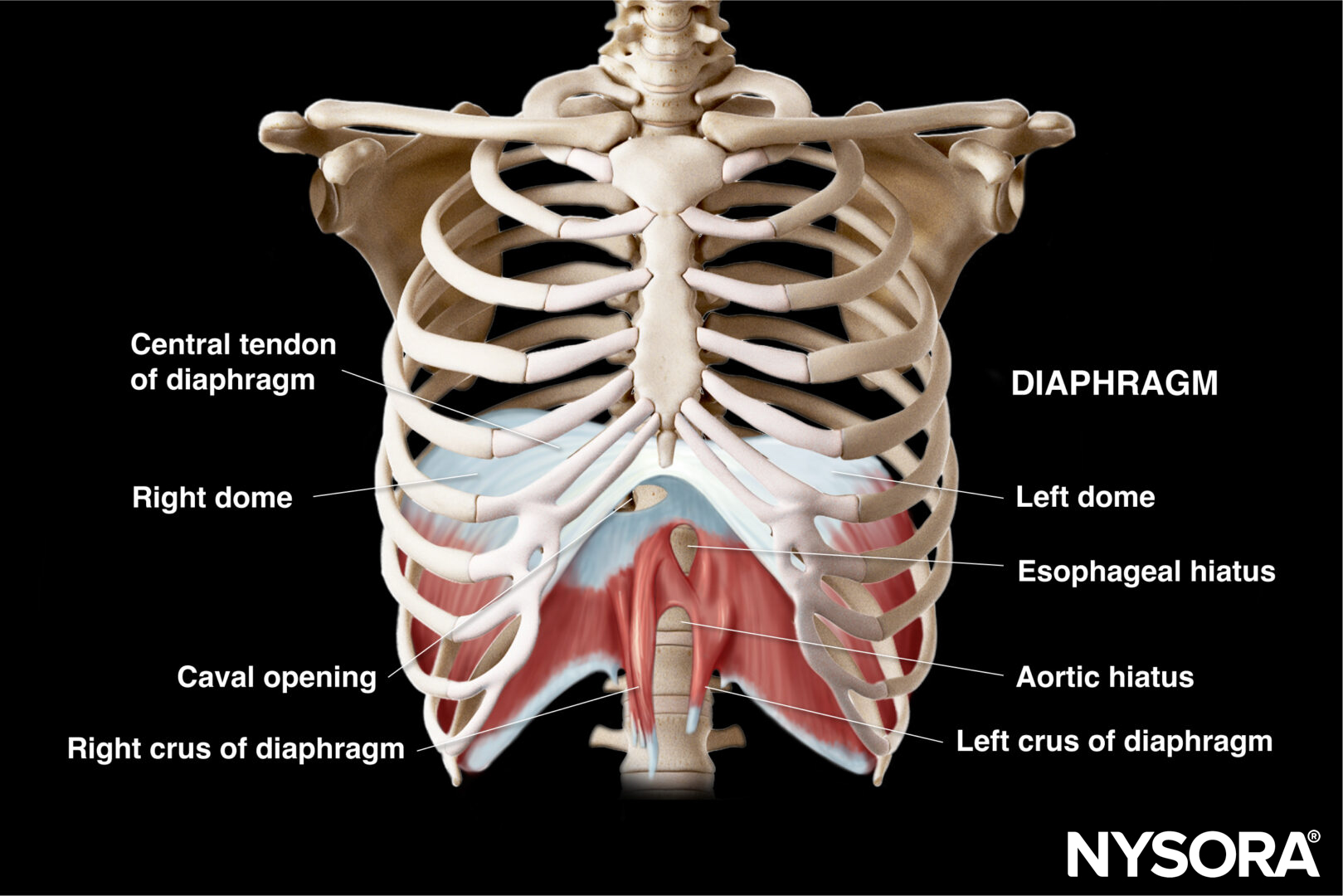
Functional anatomy of the diaphragm.
Here are the key aspects of the functional anatomy of the diaphragm:
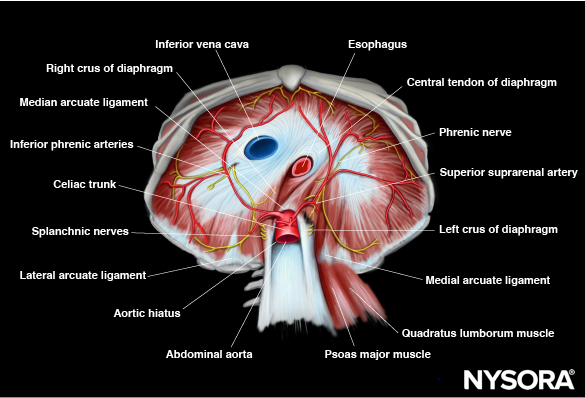
Abdominal surface of the diaphragm.
Muscle structure
The diaphragm is a large, thin, sheet-like muscle that spans the inferior thoracic aperture. It consists of two main parts: the peripheral muscle (muscular portion) and the central tendon (aponeurotic portion). The muscles of the diaphragm consist mostly (55%) of type I fibers which are slow-twitch fibers with high fatigue resistance, ensuring sustained, continuous, uninterrupted rhythmic breathing.
Attachments
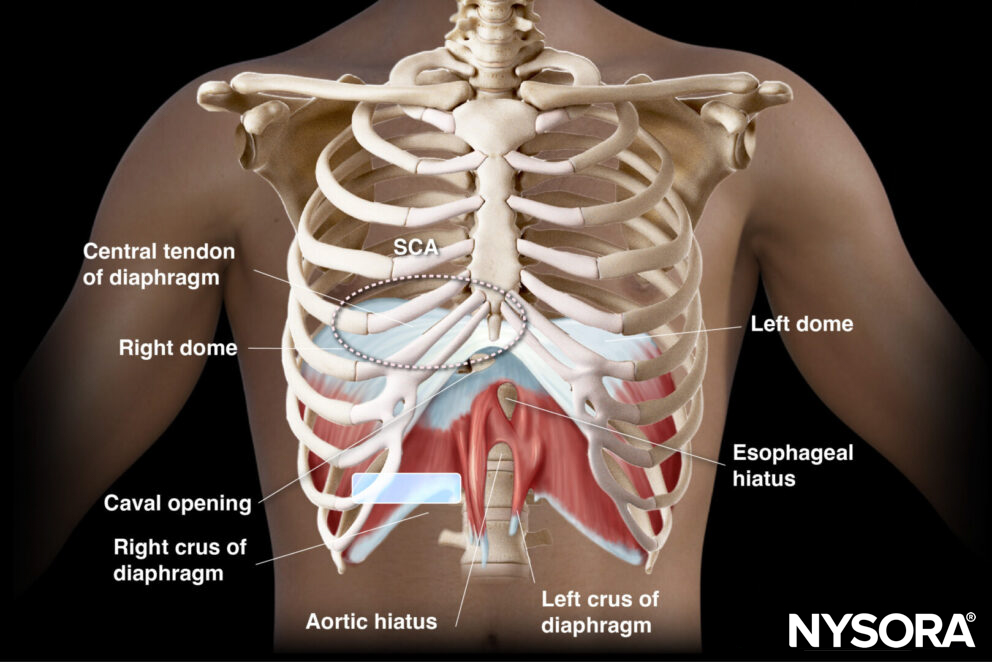
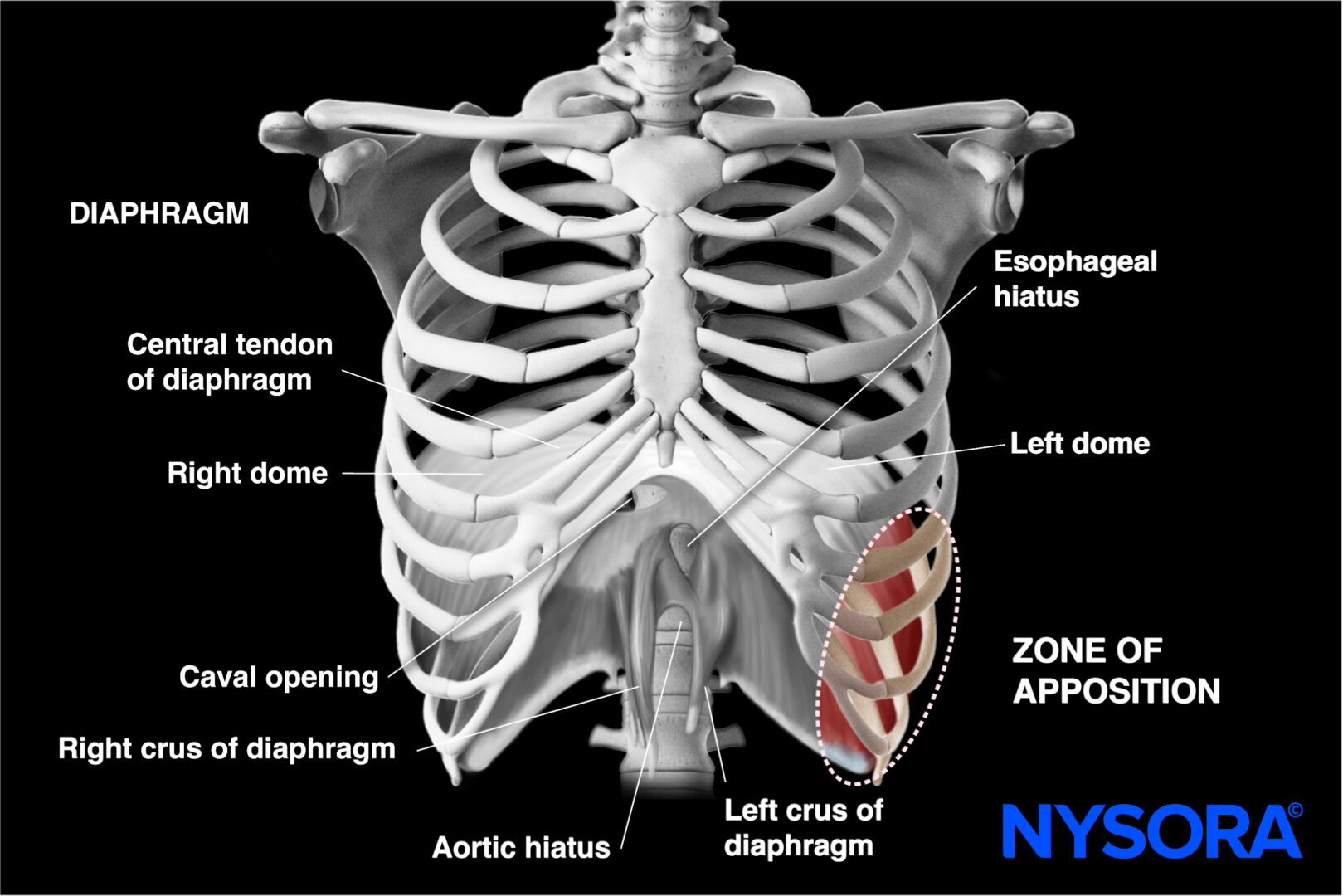
The diaphragm attaches to the lower ribs, the sternum, and the lumbar vertebrae. Peripheral attachments include the xiphoid process, costal cartilages of the lower six ribs, and the lumbar vertebrae (specifically, the L1 to L3 vertebrae). The area where the muscle attaches to the lateral thoracic wall is called the zone of apposition (ZOA).
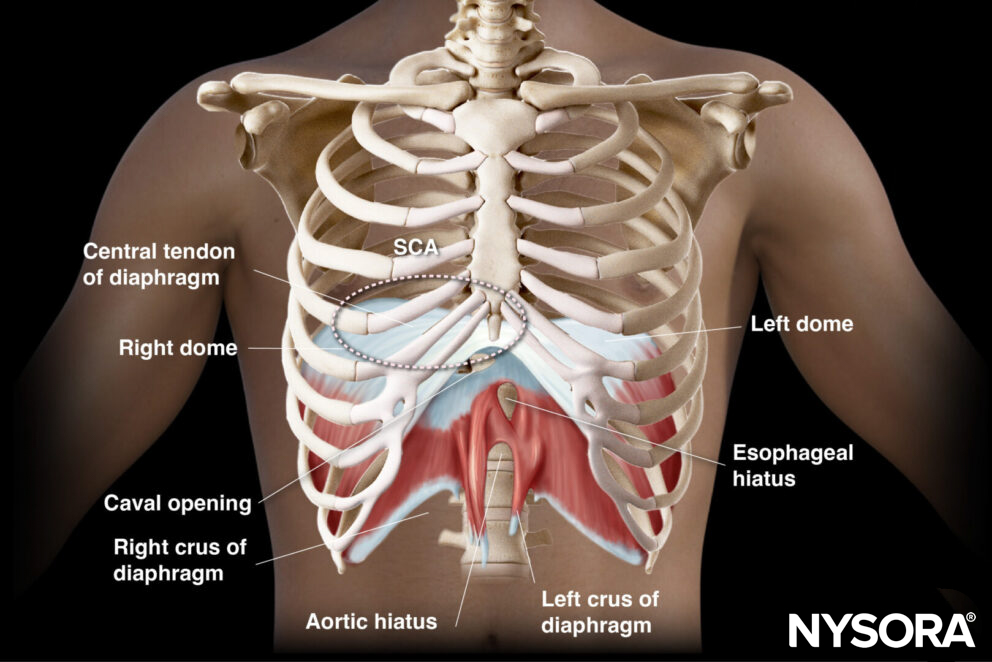
Central tendon
The central tendon is a thick aponeurotic structure at the center of the diaphragm. It gives rise to the right and left domes of the diaphragm. It lacks contractile fibers and serves as a point of attachment for the muscular fibers.
Openings
The diaphragm contains several openings for the passage of structures between the thoracic and abdominal cavities. The most significant opening is the esophageal hiatus, through which the esophagus and the vagus nerve pass. Other openings include the aortic hiatus (for the aorta, thoracic duct, and azygos vein) and the caval opening (for the inferior vena cava).
Innervation
The innervation of the diaphragm is provided by the phrenic nerves, which originate from the cervical plexus (C3-5). The phrenic nerves are essential for the motor function of the diaphragm and play a key role in the initiation and control of the breathing process.
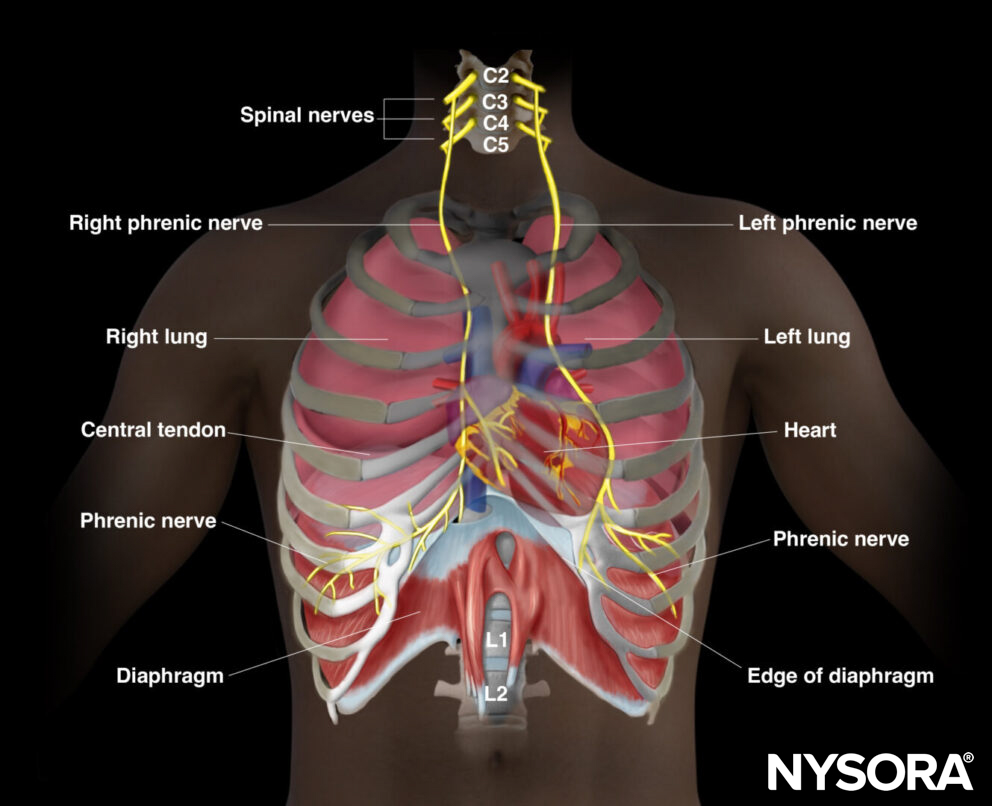
Innervation of the diaphragm.
Blood supply
Blood supply to the diaphragm comes from various arteries, including the superior and inferior phrenic arteries, branches of the thoracic and abdominal aorta, and the musculophrenic artery, which branch of the internal thoracic arteries. The diaphragm facilitates blood drainage into the brachiocephalic veins, the azygos veins, as well as the veins that converge into the inferior vena cava and the left suprarenal vein.
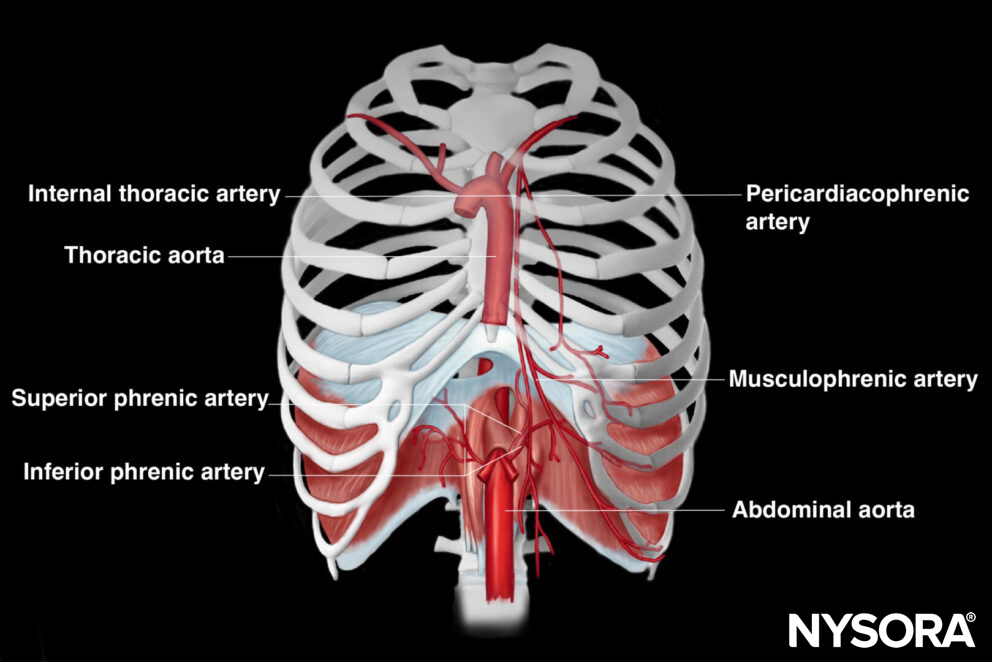
Arterial blood supply to the diaphragm.
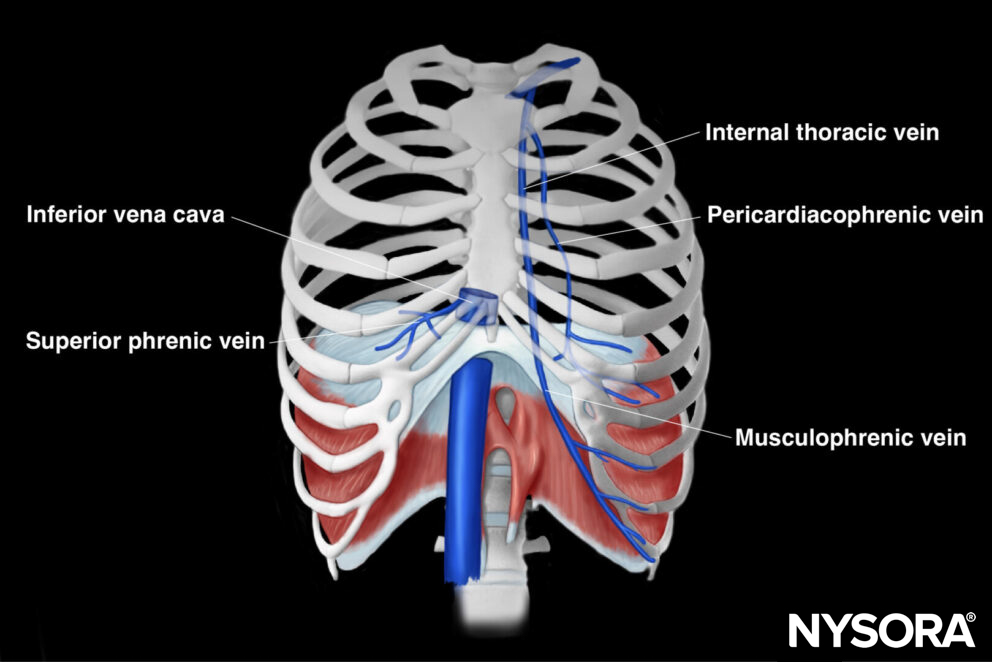
Venous drainage from the diaphragm.
Mechanism of action
During shallow breathing, diaphragmatic contraction lowers the central dome of the muscle, which leads to an increase in the vertical dimension of the thoracic cavity, resulting in decreased pleural pressur,e allowing air to flow into the lungs. Simultaneously, the descent of the central dome increases abdominal pressure, causing the anterior abdominal wall to move outward. The costal part of the diaphragm’s muscle fibers lifts the lower rib cage, generating forward (pump-handle) and outward (bucket-handle) movements. This contraction causes the diaphragm to move caudally, expanding the craniocaudal dimension of the thoracic cavity and creating negative intrathoracic pressure, facilitating lung inflation.
Accessory muscles
The diaphragm works in coordination with other respiratory muscles, such as the intercostal muscles and accessory muscles of the neck and abdomen, to facilitate breathing, especially during increased respiratory demand.
Ultrasound machine setup
Transducer:
- A curvilinear transducer (or phased array) will allow you to visualize deeper structures, such as the craniocaudal displacement of the dome of the diaphragm. The scanning of the dome of the diaphragm is usually performed in the subcostal area (SCA).
- A linear transducer will allow you to image the thickness and shortening of the muscle of the diaphragm. This is usually performed on the lateral side of the chest and is also called the zone of apposition (ZOA).
Ultrasound preset: abdominal
Orientation:
- Index mark transverse for the SCA and toward the head of the patient for the ZOA
Depth:
- 12-18 cm in the SCA; 1.5-3 cm in the ZOA
- In the SCA: use 12-18 cm (curvilinear or phased array transducer)
- In the ZOA: use 1.5-3 cm (linear transducer)
Tips
- Use M-mode in the SCA for evaluating craniocaudal displacement.
- Use B-mode in the ZOA for measuring diaphragm thickness and basic imaging.
Patient position
- Position the patient supine with both arms at their sides. This gives less variability and better reproducibility.
- Breathing instructions in awake patients include asking for normal quiet shallow breathing, deep breathing at maximal inspiration, and sniffing maneuvers.
Patient position for diaphragm ultrasound.
Tip
Slight lateral decubitus or a slight upright position is also possible and can improve access to specific regions of the diaphragm, yet reproducibility might be lower.
Landmarks
Key landmarks to identify:
- Clavicle: midclavicular line
- Axilla: anterior axillary line
- Costal margin
- Xiphoid
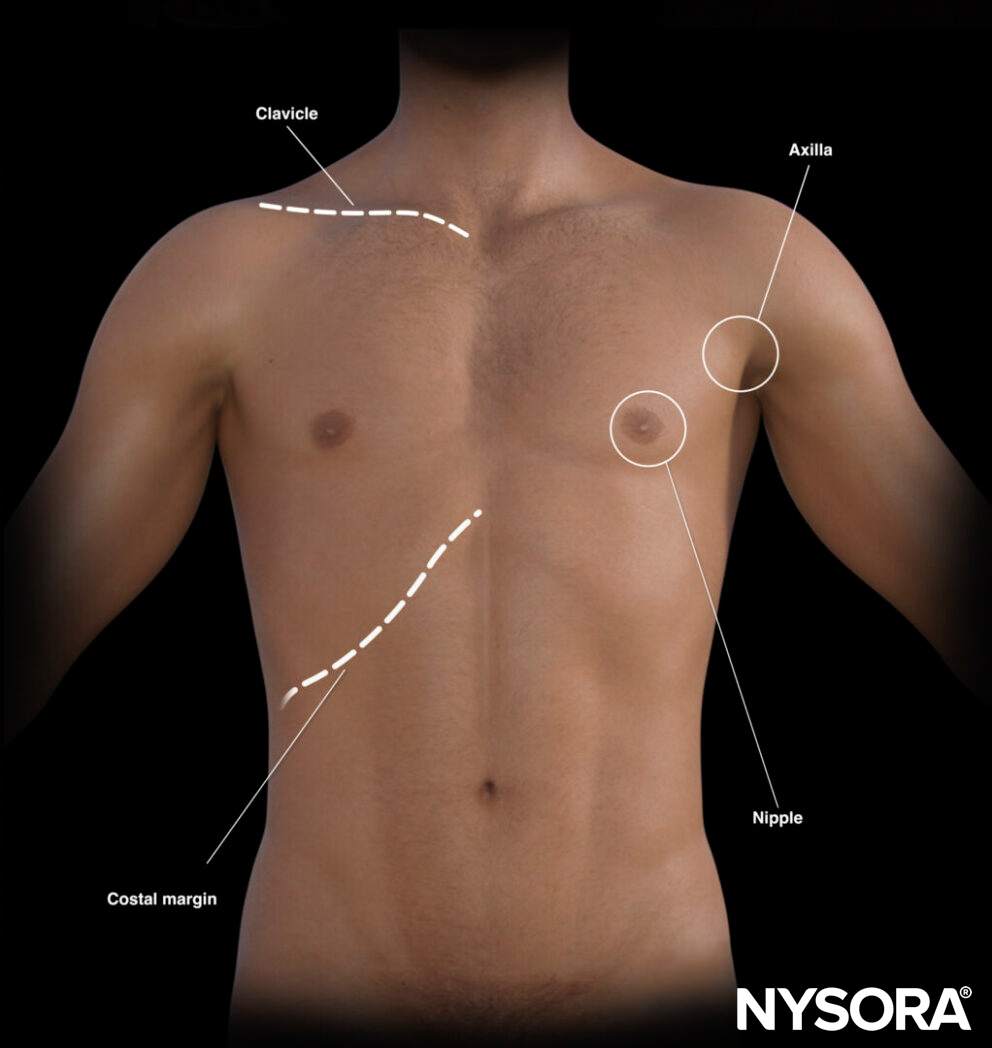
External landmarks for diaphragm assessment.
The landmarks on the thorax can also help with virtual lines for transducer placement.
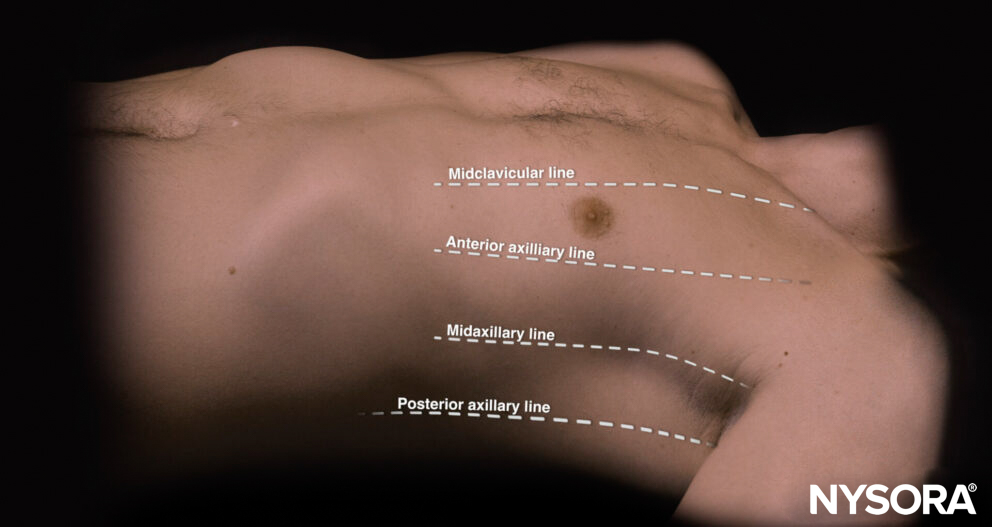
Virtual lines on the thorax.
Position the hands parallel to the clavicle with the little finger just next to the clavicle. The thumbs do not count, so remove them from the estimation. The “phrenic line” is the transverse line that follows the little finger of the lower hand. This line correlates with the end of the lung or the diaphragm.
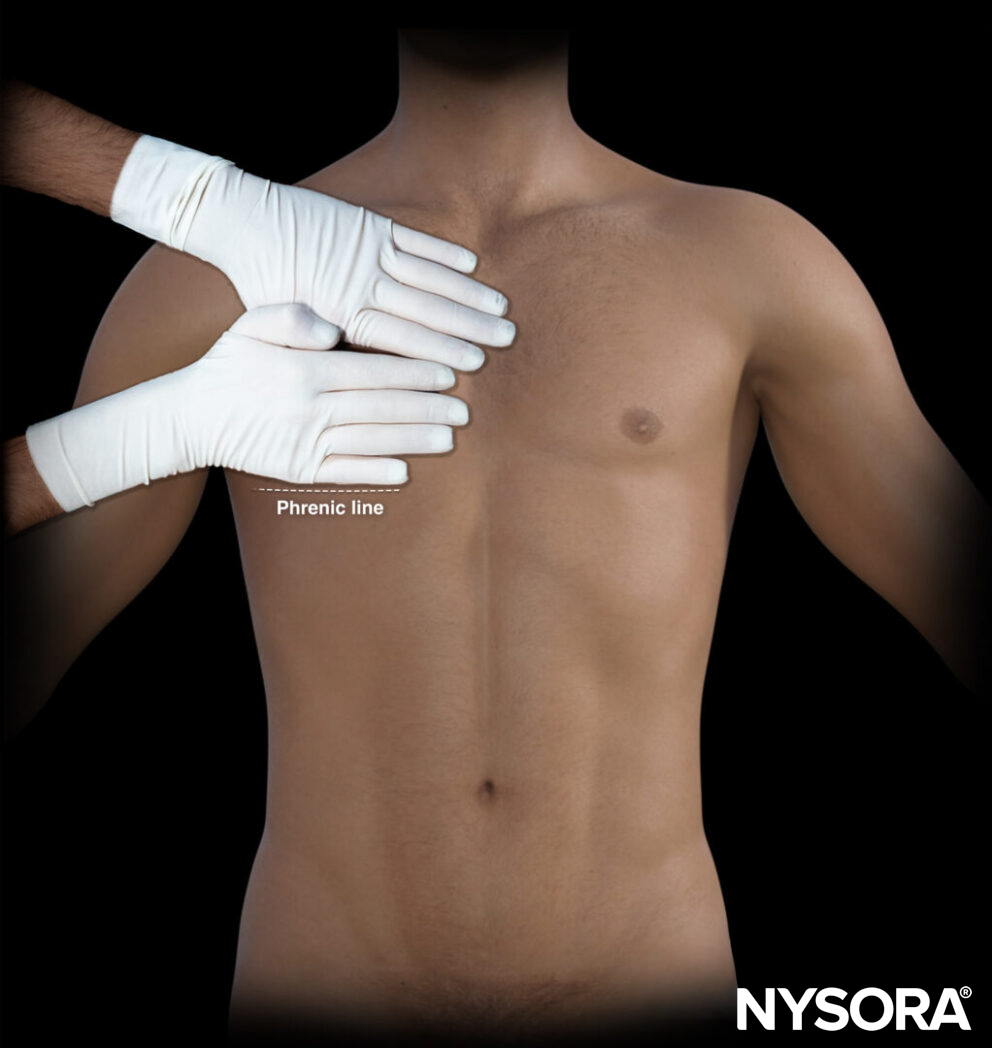
Position the hands parallel to the clavicle, little finger adjacent to it, excluding thumbs. The “phrenic line” follows the lower hand’s little finger, indicating the lung or diaphragm end.
Transducer position
Let’s review the two windows in which the diaphragm can be visualized and investigated: the subcostal area (SCA) and the zone of apposition (ZOA).
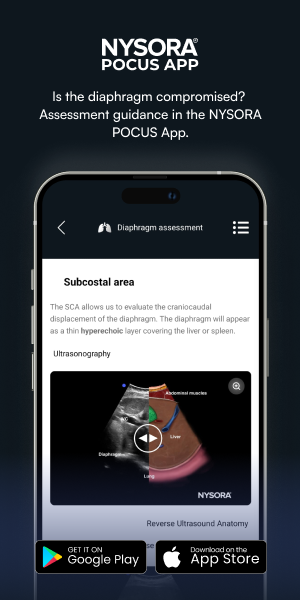
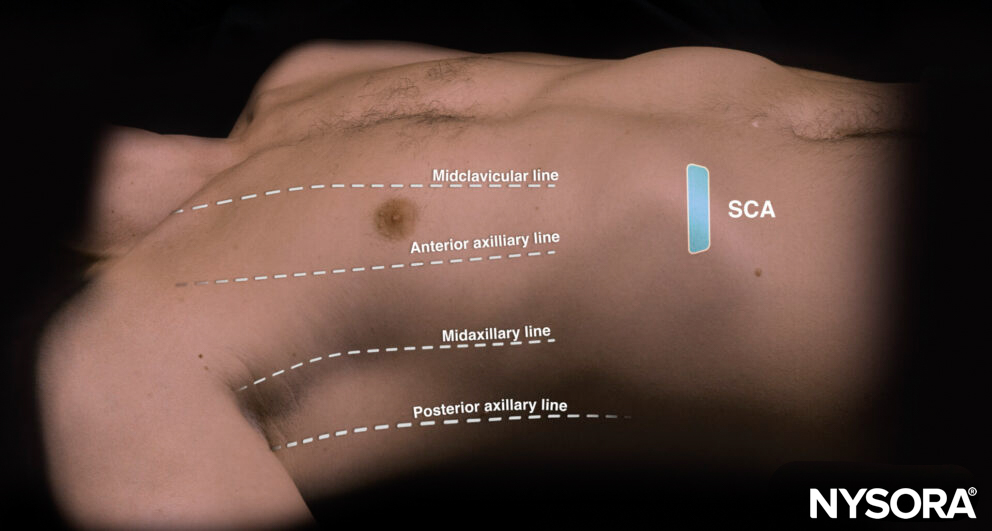
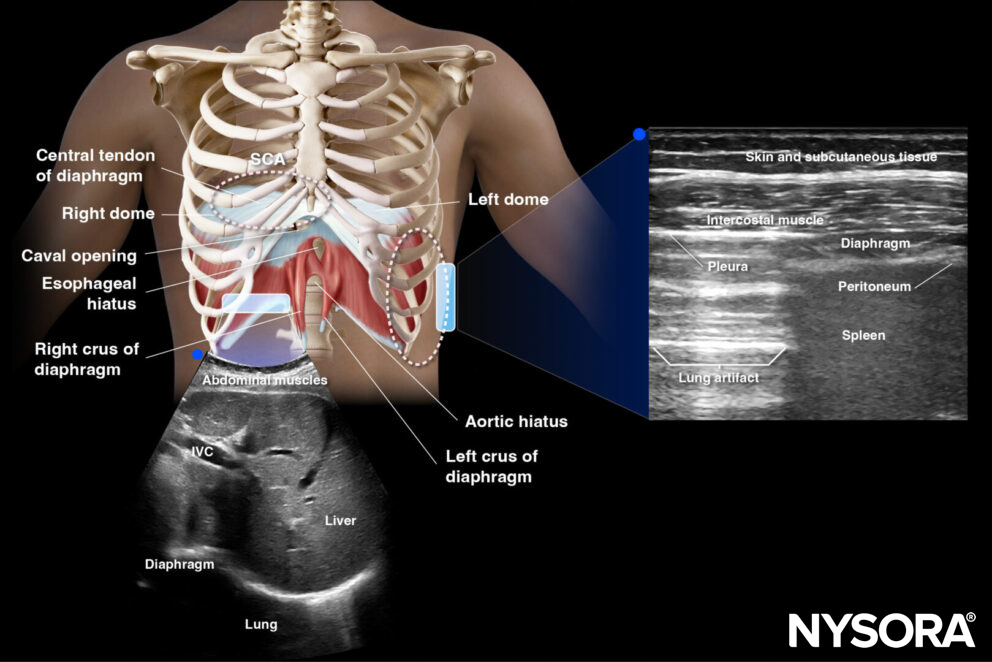
Subcostal area (SCA)
The SCA refers to the central tendinous portion of the diaphragm that forms the dome of the diaphragm.
Location of the subcostal area (SCA).
Place the transducer in a transverse position in the subcostal region on the midclavicular line, oriented to the patient’s right side. Both the liver and the spleen can serve as acoustic windows for improved visualization of the diaphragm.
Transducer position for diaphragm assessment in the subcostal area (SCA).
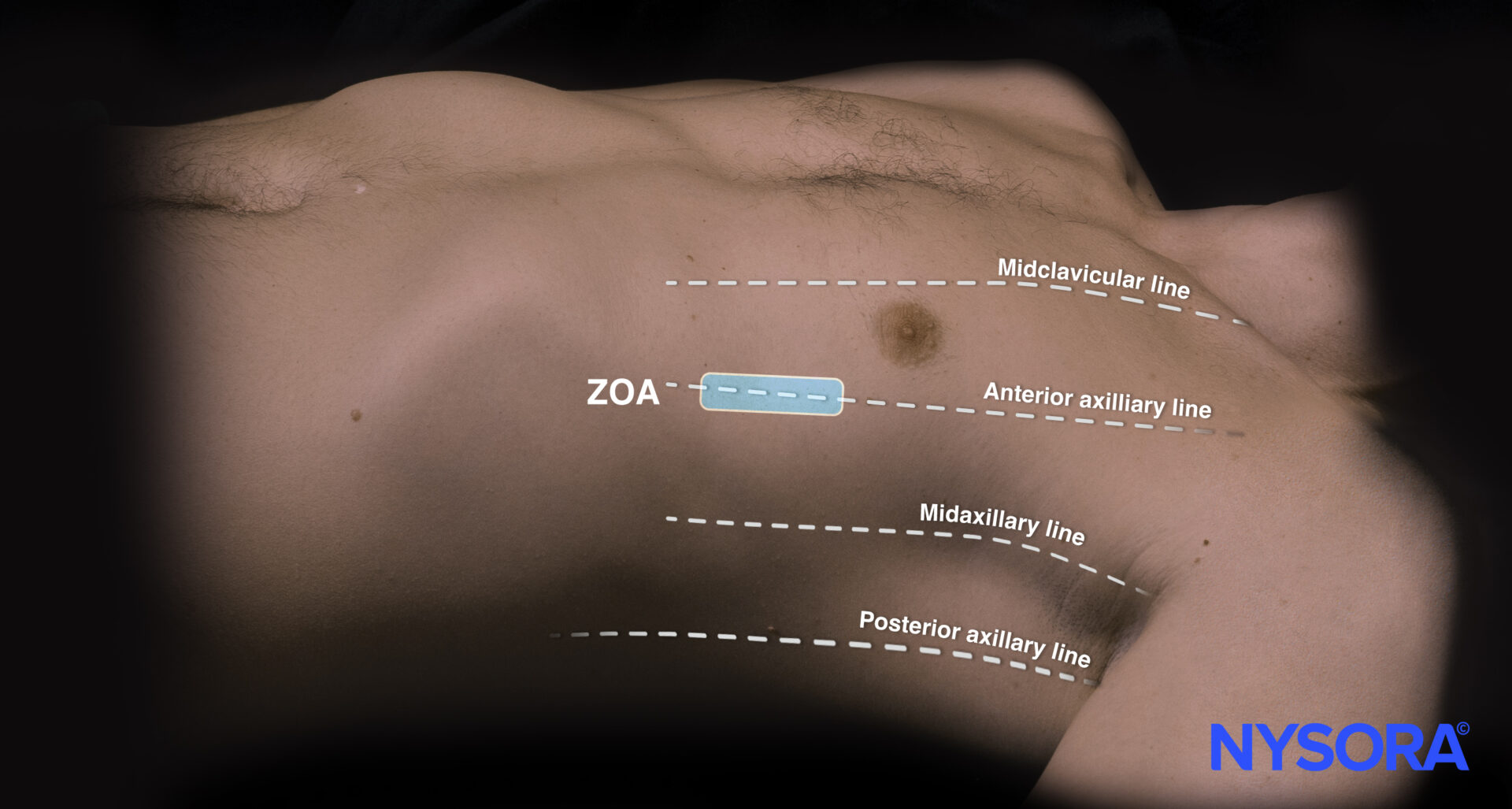
Zone of apposition (ZOA)
The ZOA refers to the area on the lateral thorax where the muscular fibers of the diaphragm run parallel and closely adhere to the inner surface of the thoracic wall.
Location of the zone of apposition.
Place the transducer parallel to the ribs between approximately the 8th and 10th intercostal space along the anterior axillary line. Visualize the pleural line cranially.
Transducer position for diaphragm assessment in the zone of apposition.
Scanning
Subcostal area (SCA)
The SCA allows us to evaluate the craniocaudal displacement of the diaphragm. The diaphragm will appear as a thin hyperechoic layer covering the liver or spleen.
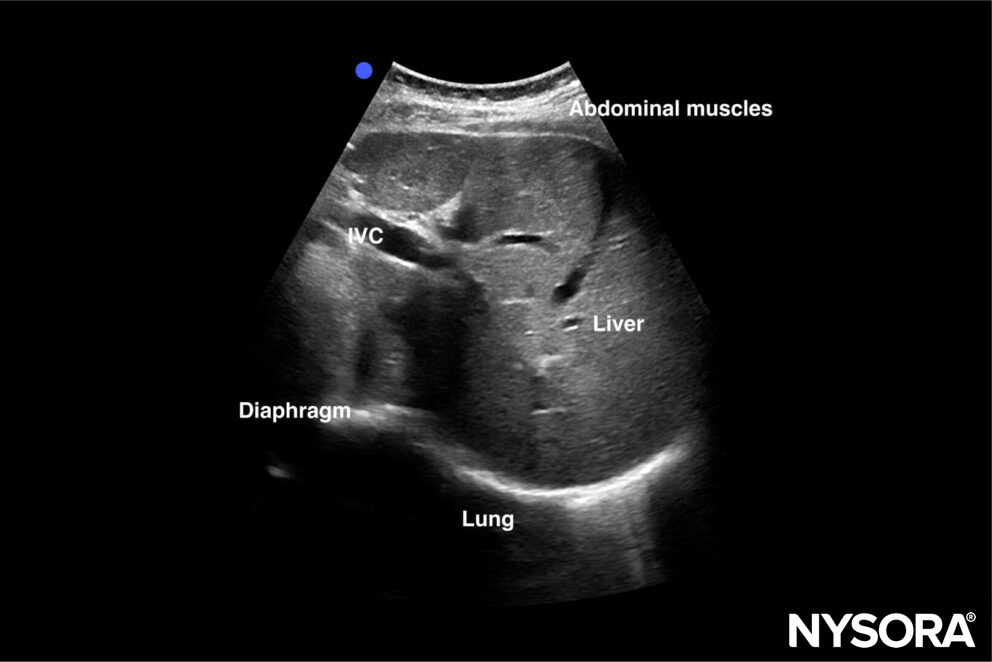
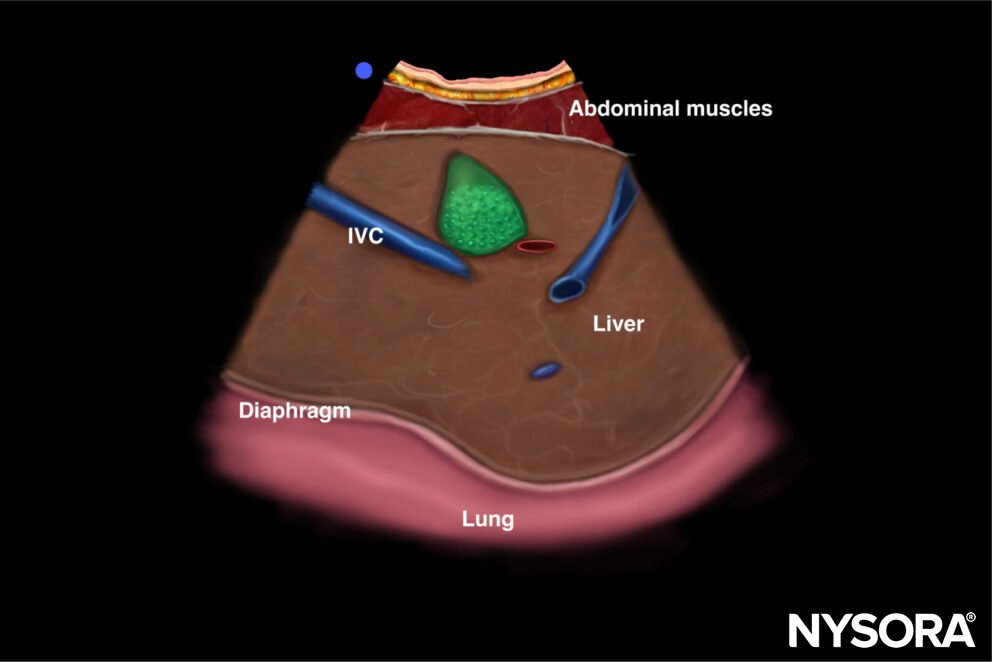
Ultrasound and Reverse Ultrasound Anatomy of the diaphragm in the subcostal area.
Note
Alternatively, a longitudinal approach can be used with the same ultrasound position.
Zone of apposition (ZOA)
The ZOA will appear as a three-layered structure immediately caudal to the pleural line. These three layers consist of two hyperechoic outer layers (i.e., the peritoneum and the pleura) and one hypoechoic inner layer (diaphragm muscle). In the ZOA, diaphragmatic thickening and shortening can be evaluated.
Note
On the left hemithorax, it can sometimes be difficult to obtain images due to the spleen’s small acoustic window, especially during deep breathing.
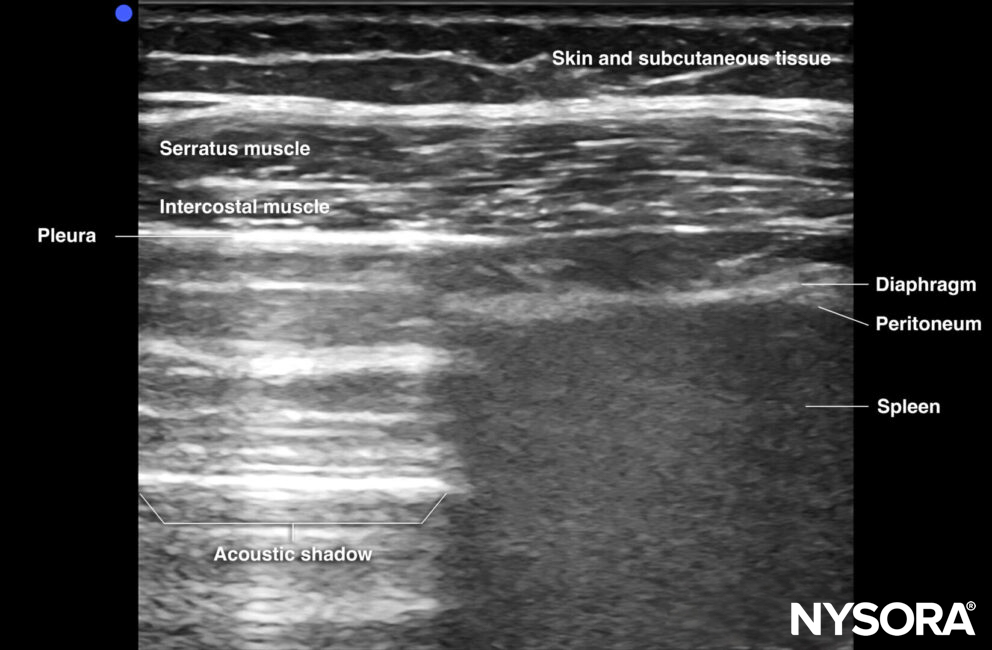
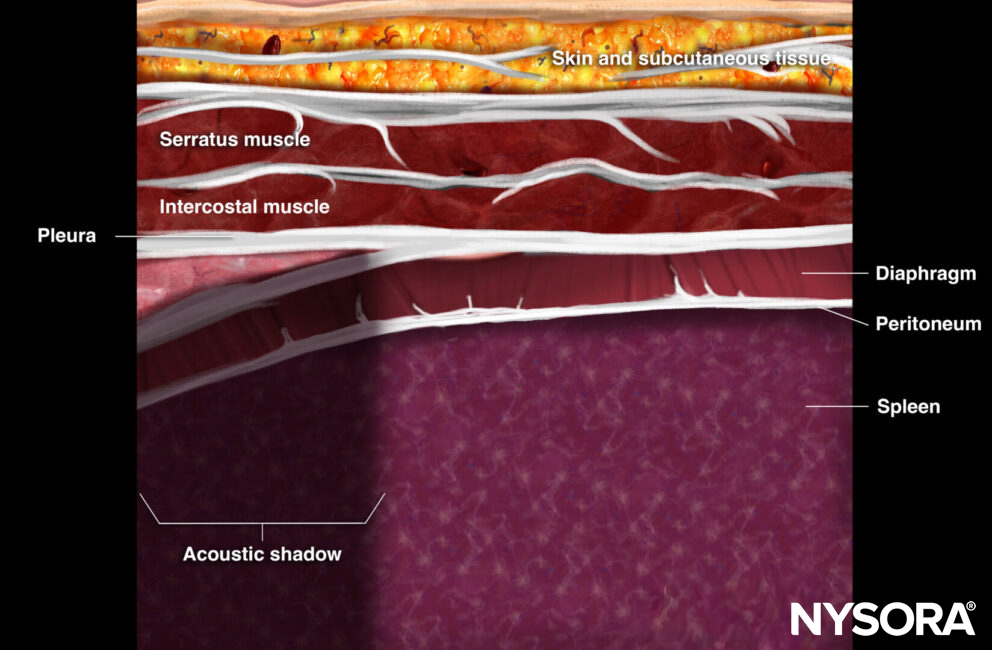
Ultrasound and Reverse Ultrasound Anatomy of the diaphragm at the zone of apposition.
Interpretation
Excursion of the diaphragm
Craniocaudal displacement or diaphragm excursions can be evaluated in the SCA window during the respiratory cycle.
Note
- Always use a curvilinear transducer and set the depth to approximately 12-18 cm. For obese patients, even deeper settings may be needed.
- The diaphragm is first identified in B-mode as a thin hyperechoic layer covering either the liver or spleen. Thereafter, M-mode is used to identify the excursion over time.

Excursion of the diaphragm in M-mode: The distance between the diaphragm’s position at inspiration (Din) and expiration (Dex) is measured.
The diaphragm is usually evaluated at three different times: during quiet normal breathing, during deep inspiration, and during a sniffing maneuver. Diaphragm excursion can be assessed by:
- Eyeballing the diaphragm in M-mode. It should look like a sinusoid during respiration in healthy patients. During inspiration, the diaphragm contracts, and during expiration it expands, resulting in a shorter and longer distance towards the ultrasound transducer. More excursion is expected during sniffing or deep breathing.
- Measuring the excursion. The following table illustrates what a normal diaphragm excursion should look like in healthy adults during different types of respiration. Tidal excursions less than these values are suggestive of diaphragmatic dysfunction.

Diaphragm dysfunction or paresis, however, results in the absence of excursion over time.
Tips
- Excursion values below 1.1-1.4 cm at the end of a spontaneous breathing trial are specific and sensitive for predicting weaning failure.
- A paradoxical movement of the diaphragm, meaning cranially on inspiration, is also highly suggestive of diaphragmatic dysfunction.
Thickening of the diaphragm
The thickness of the diaphragm can be assessed in the ZOA by comparing the thickness during in- and expiration.
Use M-mode for visualizing the diaphragm over time.
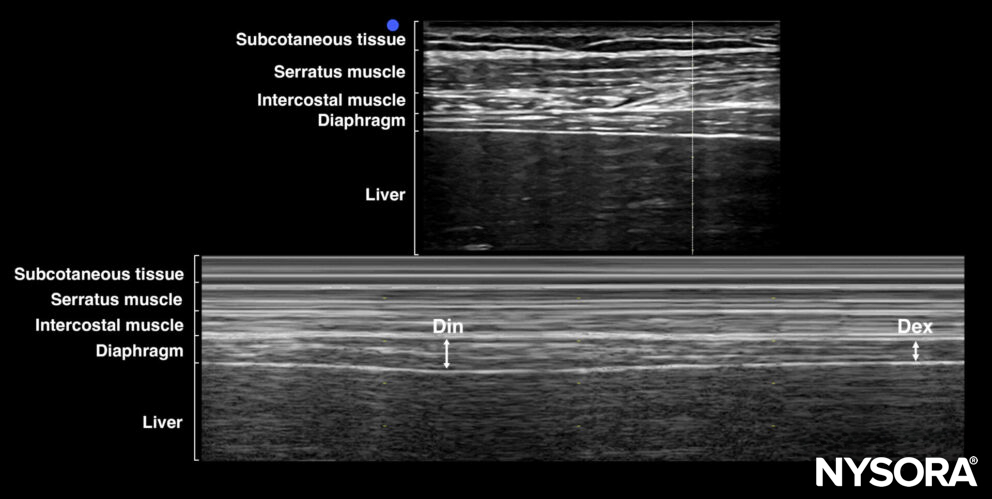
Visualization of diaphragm thickening using M-mode. Din, diaphragm thickness on end-inspiration; Dex, diaphragm thickness on end-expiration.
Thickening assessment can be performed by:
- Eyeballing the difference in thickness of the diaphragm during in- and expiration.
- Comparing the thickness of the diaphragm at shallow breathing with a breath-holding maneuver.
- Normal diaphragm thickness in healthy individuals at the ZOA is 1.7mm +/- 0.2 mm in shallow breathing, while increasing to 4.5 +/- 0.9 mm when doing a breath-holding maneuver at total lung capacity (TLC).
- Calculating the thickening fraction
Note
This measurement is the least reliable of all the mentioned techniques. Since the diaphragm is only a few millimeters thick, only a small margin of error can result in very different measurement outcomes, and this results in high interobserver variability.
- The thickening fraction (TF), measured at the ZOA, is defined as the thickness of the diaphragm on end-inspiration (Din) minus the thickness of the diaphragm on end-expiration (Dex) divided by Dex.
TF = (Din – Dex)/Dex
A thickening fraction of less than 20% is suggestive of diaphragmatic dysfunction.
Tip
A TF less than 30-36% at the end of a spontaneous breathing trial is sensitive and specific for predicting weaning failure.
Diaphragm dysfunction or paresis will show no thickening in B-mode or M-mode
Notes
- Excursion of the diaphragm can also be assessed in the ZOA. Identify and mark the highest point on the skin where the ZOA was observable against the rib cage during both end-inspiration and end-expiration. Assess the distance between the two skin markings. No normal values are available yet, but in the presence of diaphragm paralysis, no excursion will be present.
- This technique has been shown to be similarly successful in the right hemithorax as the SCA scanning technique (100% vs 98.7%). However, on the left hemithorax, this technique proved more consistent than the SCA scanning technique (100% vs 34%) due to the usually small acoustic window of the spleen.
Overview
Clinical updates
Sagmal et al. (Heart & Lung, 2026) conducted a prospective ED study of 77 patients with acute respiratory failure requiring NIV and found that right diaphragm excursion <1.4 cm (measured by M-mode POCUS immediately before NIV) predicted NIV failure with an AUC of 0.86, sensitivity 85.7%, specificity 85.7%, NPV 94%, and overall accuracy 84.4%. In multivariable analysis, excursion <1.4 cm was the strongest independent predictor of NIV failure (OR 42.12), along with older age and more negative base excess, and was associated with markedly higher intubation rates (66.7% vs 6.0%), supporting early diaphragm ultrasound as a risk-stratification tool in the ED.
- Sagmal YE, Eksioglu M, Kaymak BA, Ozturk TC. Diaphragm excursion as a predictor of noninvasive ventilation failure in the emergency department: A prospective study. Heart Lung. 2026;75:313-320.