Learning objectives
- Describe the physiological changes associated with anorexia nervosa
- Manage anorexia nervosa patients in the perioperative period
Definition
- Anorexia nervosa is a psychiatric disorder characterized by an intense fear of gaining weight and a distorted perception of weight, resulting in an abnormally low body weight
Physiological changes
| System | Changes |
|---|---|
| Cardiovascular | Hypotension, bradycardia, mitral valve prolapse, impaired myocardial contractility, cardiomyopathy, ECG changes (risk of arrhythmias) |
| Respiratory | Metabolic alkalosis, decreased lung compliance, aspiration pneumonia, pneumothorax, and pneumomediastinum |
| Renal | Proteinuria, reduced glomerular filtration rate, ↓Na+, ↓K+, ↓Mg2+, ↓Cl−, ↓H+, ↓Ca2+, ↓PO42− hypophosphatemia, renal calculi |
| Gastrointestinal | Enlarged salivary glands, dental caries, periodontitis, Mallory–Weiss tears, esophageal strictures, esophagitis, gastritis, gastric dilatation/infarction/perforation, ↑ gastric emptying time, risk of refeeding syndrome, fatty liver, hepatomegaly, cirrhosis, ↑amylase, abnormal liver function tests |
| Endocrine | FSH, ↓LH, ↓GnRH, ↑cortisol, ↑GH, ↓glucose, ↓insulin ↓T3, ↓T4, ↔TSH, impaired thermoregulation |
| Immunological | Leucopenia, thrombocytopenia, hemolytic anemia |
| Hematological | Bone marrow hypoplasia |
| Neurological | White and gray matter changes, ↓cognitive function, coma EEG abnormalities, seizures, neuropathy, ↑pain threshold |
| Musculoskeletal | Myalgia, myopathy, rhabdomyolysis, osteopenia, osteoporosis, stress fractures |
| Dermatological | Laguno hair, Russell’s sign, poor wound healing |
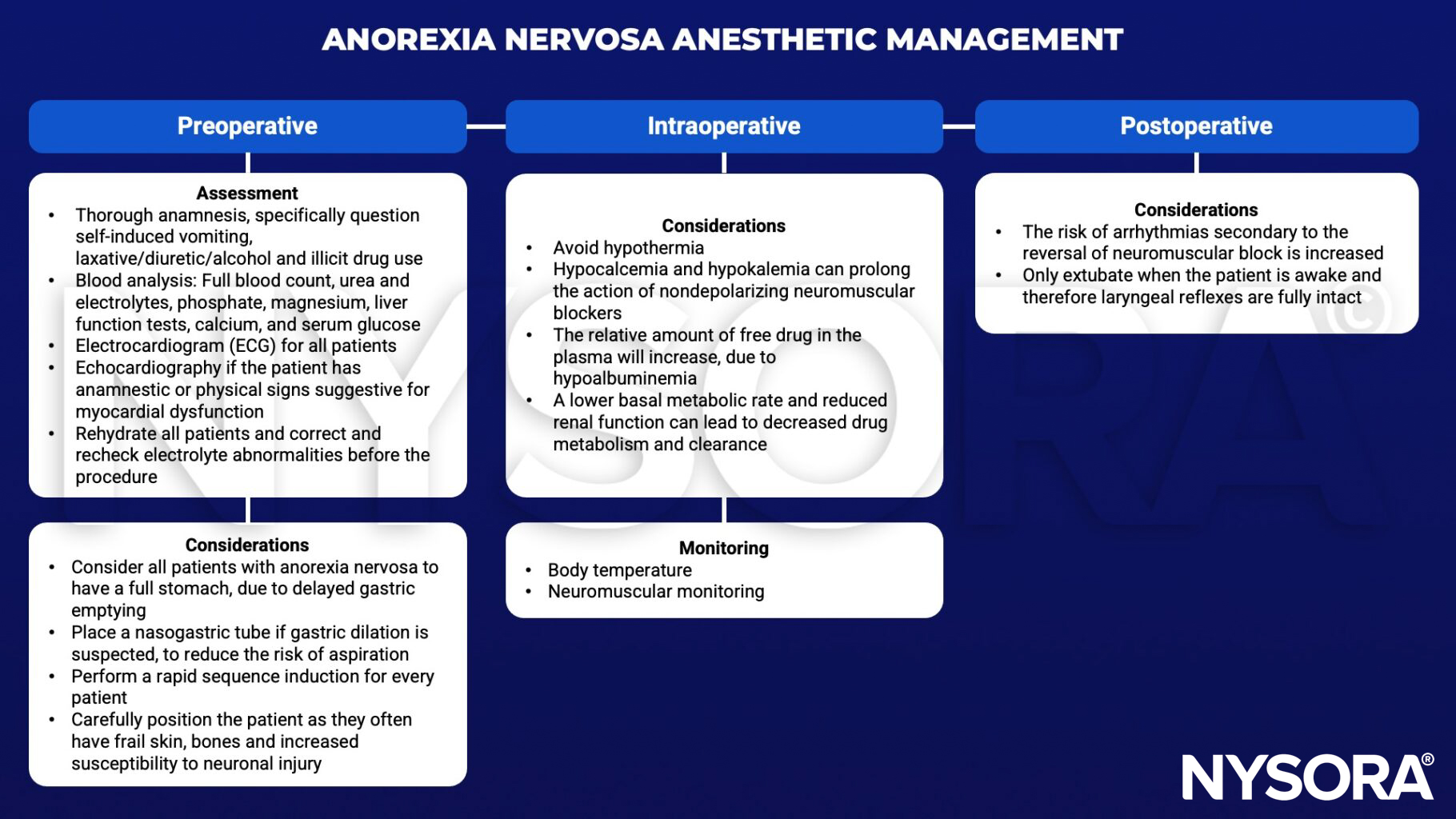
Management
Suggested reading
- Denner AM, Townley SA. Anorexia nervosa: perioperative implications. Continuing Education in Anaesthesia Critical Care & Pain. 2009;9(2):61-4.
Clinical updates
Trapani and Rubino (Pediatrics, 2025) provide an updated review of acute and chronic medical complications of anorexia nervosa in children and adolescents, organized by body system, highlighting that up to 20% of hospitalized youth exhibit medical instability (eg, bradycardia, hypotension, electrolyte derangements) and that cardiovascular causes account for ~60% of AN-related deaths. They emphasize that many complications, such as reduced left ventricular mass, pericardial effusion, hypogonadotropic hypogonadism, low-T3 syndrome, renal impairment, and refeeding syndrome are largely reversible with careful weight restoration, whereas low bone mineral density and neurocognitive deficits may be only partially reversible, underscoring the need for early multidisciplinary intervention and cautious refeeding protocols.
Dahlgren et al. (Journal of Eating Disorders, 2025) report the first European case of structured ketamine-assisted psychotherapy (KAP) for anorexia nervosa, describing a woman in her late 20s with AN and comorbid depression who received four intramuscular ketamine sessions within an ACT framework followed by three boosters, showing rapid reductions in ED psychopathology, depression, and anxiety sustained at 5 months. Symptom improvement was greatest during KAP with psychotherapy, partially worsened during the therapy-free interval, and improved again after boosters. Ketamine was well tolerated, with no emergent adverse effects; however, weight declined by follow-up, underscoring the need for structured post-treatment monitoring and integration support.
van den Berg et al. (BJA Education, 2023) provide a systems-based perioperative framework for patients with anorexia nervosa, emphasizing contraindications to elective surgery with bradycardia <40 beats·min⁻¹ or systolic BP <80 mmHg, overestimation of GFR from low creatinine, risk of aspiration from delayed gastric emptying/SMAS, and avoidance of depolarizing neuromuscular blockers with preference for sugammadex reversal. They highlight altered PK/PD in BMI <18.5 kg·m⁻² (risk of propofol under- or overdosing; TCI models not validated), recommend EEG-guided titration and lung-protective, carefully balanced fluid strategies to avoid heart failure, and stress prevention of refeeding syndrome — checking/correcting phosphate, K⁺, Mg²⁺, giving thiamine 100 mg, initiating 10–20 kcal·kg⁻¹·day⁻¹ with 33% increments, and accounting for propofol’s 1.1 kcal·mL⁻¹ caloric load.