About to perform the interscalene block?
Review transducer position, anatomy, and needle trajectory in seconds.
FACTS
- Indications: Shoulder and upper arm surgery, surgery of the clavicle (combined with cervical plexus nerve block)
- Transducer position: Transverse on the neck, 3–4 cm superior to the clavicle, over the external jugular vein
- Goal: Local anesthetic spread around superior and middle trunks of brachial plexus, between the anterior and middle scalene muscles
- Local anesthetic: 7–15 mL
GENERAL CONSIDERATIONS
US guidance allows for visualization of the spread of the local anesthetic and additional injections around the brachial plexus if needed to ensure an adequate spread of local anesthetic, improving nerve block success. The ability to visualize local anesthetic spread and to inject multiple aliquots also allows for a reduction in the volume of local anesthetic required to accomplish the nerve block.
ULTRASOUND ANATOMY
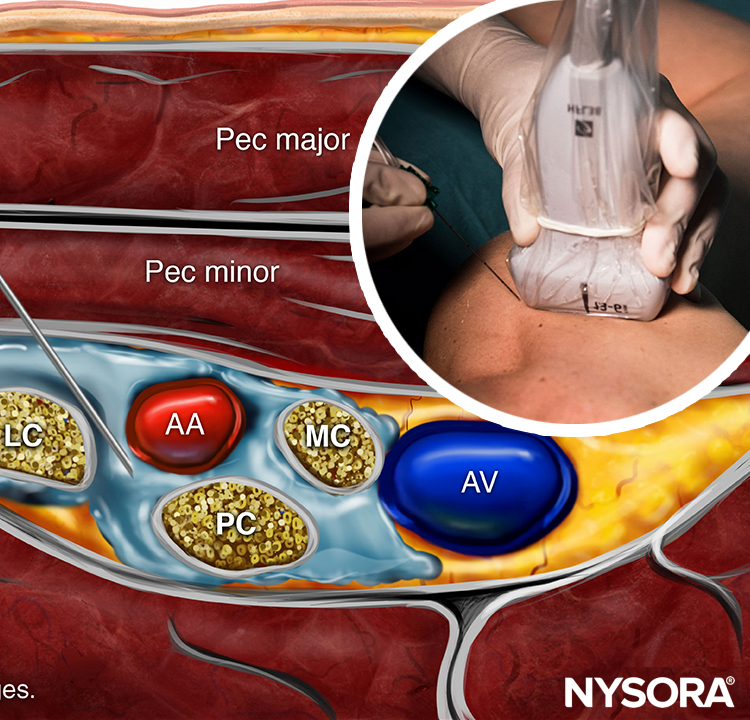
The brachial plexus at the interscalene level is seen lateral to the carotid artery and internal jugular vein, between the anterior and middle scalene muscles (Figures 1 and 2).
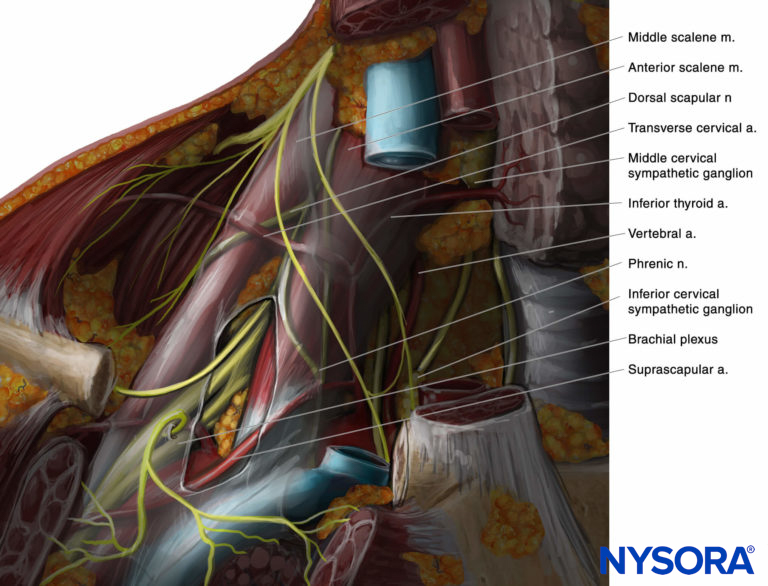
FIGURE 1. Anatomic relationships of the brachial plexus at the interscalene level
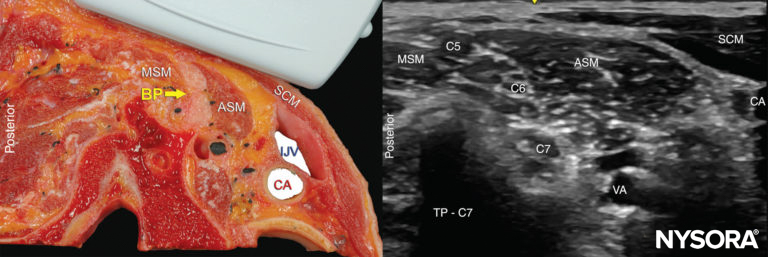
FIGURE 2. Cross-section anatomy for interscalene brachial nerve block and transducer position to obtain the desired views. The plexus (BP) is seen between the middle scalene muscle (MSM) laterally and anterior scalene muscle (ASM) medially. The ultrasound image includes a partial view of the lateral border of the sternocleidomastoid muscle (SCM) the internal jugular vein (IJV), carotid artery (CA) and the transverse process of C7 (TP-C7).
The prevertebral fascia, superficial cervical plexus, and sternocleidomastoid muscle are seen superficial to the plexus. The transducer is moved in the proximal-distal direction until two or more of the brachial plexus elements are seen in the space between the scalene muscles. Depending on the depth of field selected and the level at which scanning is performed, the first rib and/or the apex of the lung may be seen. The brachial plexus is typically visualized at a depth of 1–3 cm.
BLOCK DISTRIBUTION
The interscalene approach to brachial plexus block results in reliable anesthesia of the shoulder and upper arm (Figure 3). The supraclavicular branches of the cervical plexus, supplying the skin over the acromion and clavicle, are also blocked due to the proximal and superficial spread of local anesthetic. The inferior trunk (C8-T1) is usually spared unless the injection occurs at a more distal level of the brachial plexus.

FIGURE 3. Sensory distribution of the interscalene brachial plexus nerve block (in red). Ulnar nerve distribution area (C8-T1) can also be accomplished by using larger volume (e.g. 15-20 ml) and using low interscalene nerve block where the injection occurs between the ISB and supraclavicular nerve block.
EQUIPMENT
The equipment needed for an interscalene brachial plexus nerve block includes the following:
- Ultrasound machine with a linear transducer (8–14 MHz), sterile sleeve, and gel
- Standard nerve block tray
- A 20-mL syringe containing the local anesthetic
- A 5-cm, 22-gauge, short-bevel, insulated stimulating needle
- Peripheral nerve stimulator
- Opening injection pressure monitoring system
- Sterile gloves
Learn more about Equipment for Peripheral Nerve Blocks
LANDMARKS AND PATIENT POSITIONING
Any position that allows for comfortable placement of the ultrasound transducer and needle advancement is appropriate. The nerve block is typically performed with the patient in a supine, beach chair, or semi-lateral decubitus position, with the patient’s head facing away from the side to be blocked (Figure 4). The latter position may prove more ergonomic, especially during an in-plane approach from the lateral side, in which the needle enters the skin at the posterolateral aspect of the neck. A slight elevation of the head of the bed is often more comfortable for the patient and allows for better drainage and less prominence of the neck veins. The patient should be asked to reach for the ipsilateral knee in order to lower the shoulder and provide more space for the nerve block performance.
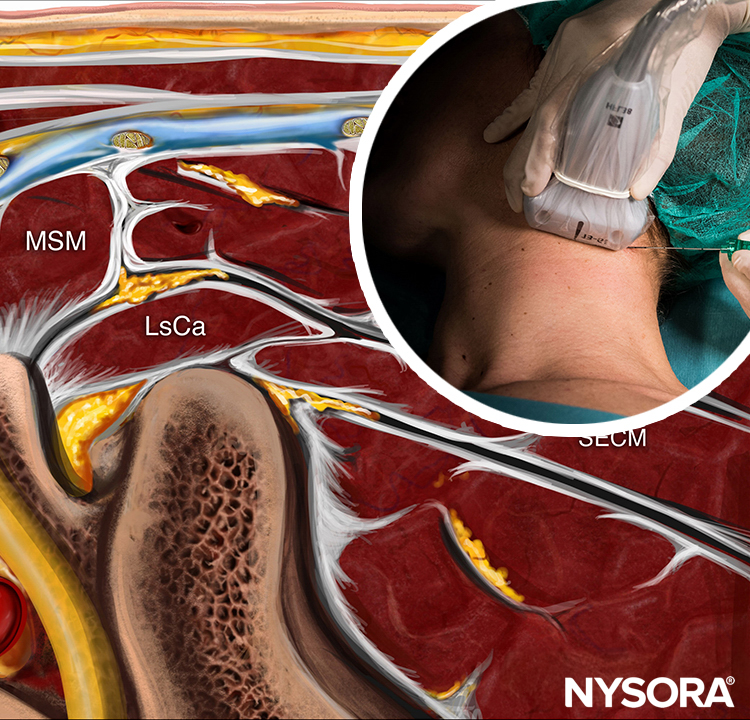
Knowledge of the underlying anatomy and the position of the brachial plexus is important to facilitate recognition of the ultrasound anatomy. Scanning usually begins just below the level of the cricoid cartilage and medial to the sternocleidomastoid muscle with the goal of identifying the carotid artery.
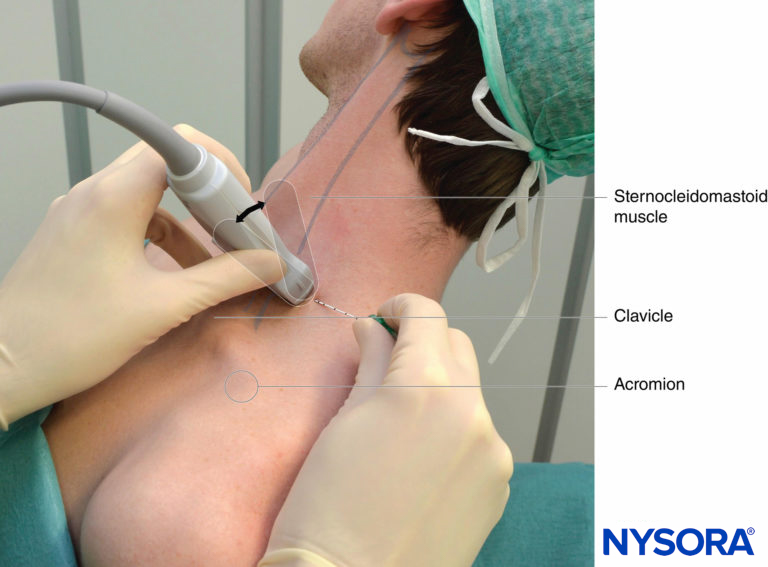
FIGURE 4. Ultrasound-guided interscalene brachial plexus nerve block: transducer and needle position to obtain the desired ultrasound image for an in-plane approach. The knowledge of external landmarks substantially facilitates and shortens the time to obtain the view necessary for nerve block performance. The transducer is positioned behind the clavicular head of the sternocleidomastoid muscle (SCM) and over the external jugular vein (not seen). The patient is in a semi-sitting position. Tilting the transducer in the caudad direction can facilitate recognition of the brachial plexus (arrow).
GOAL
The goal of this nerve block is to place the needle in the tissue space between the anterior and middle scalene muscles and inject local anesthetic until the spread around the brachial plexus is documented by ultrasound. The volume of the local anesthetic and the number of needle insertions are determined during the procedure and depending on the adequacy of the observed spread of local anesthetic.
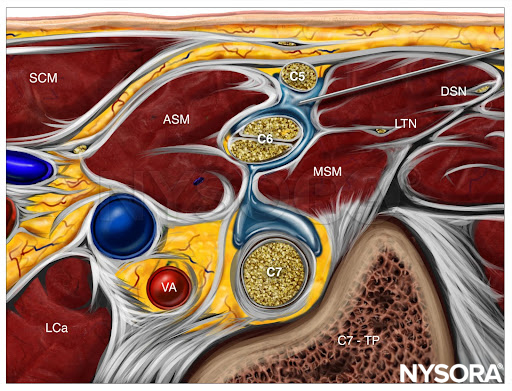
Reverse Ultrasound Anatomy for an interscalene brachial plexus block with needle insertion in-plane and local anesthetic spread (blue). SCM, sternocleidomastoid; ASM, anterior scalene muscle; LCa, longus capitis muscle; VA, vertebral artery; MSM, middle scalene muscle; LTN, long thoracic nerve; DSN, dorsal scapular nerve; C7-TP, transverse process of C7.
TECHNIQUE
With the patient in the proper position, the skin is disinfected and the transducer is positioned in the transverse plane to identify the carotid artery (Figure 5-A). Once the artery has been identified, the transducer is moved slightly laterally across the neck. The goal is to identify the anterior and middle scalene muscles and the elements of the brachial plexus that is located between them. It is recommended to use the color Doppler to identify vascular structures and avoid them. The needle is then inserted in-plane toward the brachial plexus, typically in a lateral-to-medial direction (Figure 6), although a medial-to-lateral needle orientation can also be used if there is no room for the former. The needle should always be aimed in between the roots instead of directly at them in order to minimize the risk of accidental nerve injury. As the needle passes through the prevertebral fascia, a certain “pop” is often appreciated. When nerve stimulation is used (0.5 mA, 0.1 msec), the entrance of the needle in the interscalene groove is often associated with a motor response of the shoulder, arm, or forearm as another confirmation of proper needle placement. After careful aspiration to rule out intravascular needle placement, 1–2 mL of local anesthetic is injected to verify proper needle placement (Figure 7-A). It is necessary to ensure that high resistance to injection is absent to decrease the risk of intrafascicular injection. Injection of several milliliters of local anesthetic often displaces the brachial plexus away from the needle. Additional advancement of the needle 1–2 mm toward the brachial plexus may be beneficial to ensure the proper spread of the local anesthetic (Figure 7-B). When injection of the local anesthetic does not appear to result in a spread around the brachial plexus, additional needle repositioning and injections may be necessary.
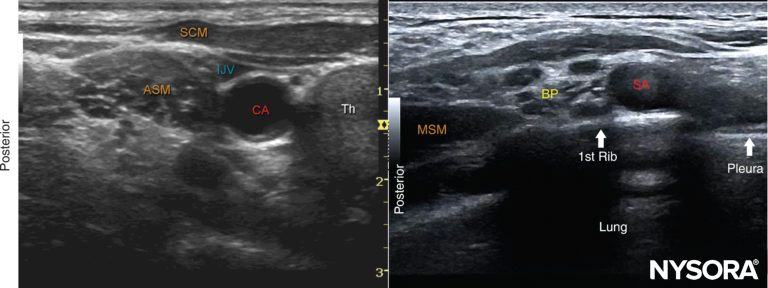
FIGURE 5. (A)Ultrasound image just below the level of the cricoid cartilage and medial to the sternocleidomastoid muscle (SCM). ASM, anterior scalene muscle; CA, carotid artery; IJV, internal jugular vein; SCM, sternocleidomastoid muscle; Th, thyroid gland. (B) View of the brachial plexus (BP) at the supraclavicular fossa. When identification of the brachial plexus at the interscalene level proves difficult, the transducer is positioned at the supraclavicular fossa to identify the BP superficial and posterior to the subclavian artery (SA). The transducer is then slowly moved cephalad while continuously visualizing the brachial plexus until the desired level is reached.
NYSORA TIPS
• When visualization of the brachial plexus between the scalene muscles proves difficult, a “traceback” technique can be used. The transducer is lowered to the supraclavicular fossa. At this position, the brachial plexus is identified posterior and superficial to the subclavian artery (Figure 5-B). From here, the brachial plexus is traced cranially to the desired level.
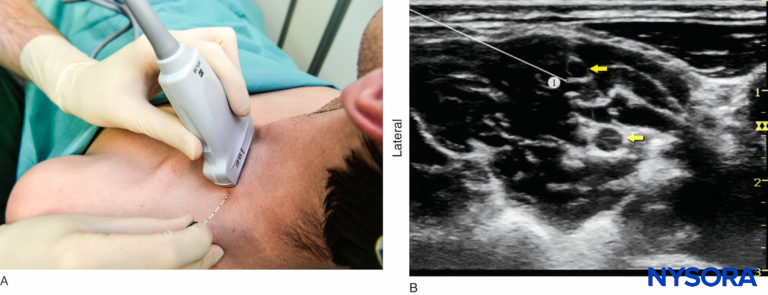
FIGURE 6. (A) Transducer placement and needle insertion. (B) Position of the needle (1) for the interscalene brachial plexus nerve block using an in-plane approach. The needle tip is seen in contact with the elements of the brachial plexus (yellow arrows); this always results in high injection pressure (> 15 psi)—indicating that the needle should be withdrawn slightly away from the trunk.
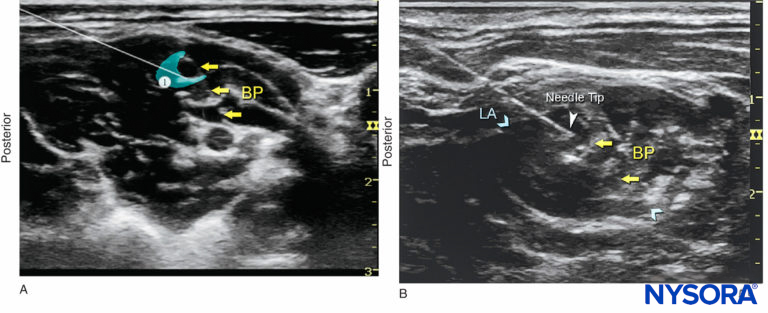
FIGURE 7. (A) A small volume of local anesthetic (blue-shaded area) is injected through the needle to confirm proper needle placement. A properly placed needle tip will result in distribution of local anesthetic between and/or alongside roots of the brachial plexus (BP). (B) An actual needle (white arrowhead) placement in the interscalene groove, with dispersion of local anesthetic (LA) (blue-shaded area or arrows) surrounding the BP.
TIPS
• It is not necessary to elicit a motor response to nerve stimulation; however, when it occurs at intensities < 0.5 mA, the needle should be slightly withdrawn before injecting as it may be intraneural.
• The neck is a highly vascular area, and care must be exercised to avoid needle placement or injection into the vascular structures. Of particular importance is to avoid the vertebral artery and branches of the thyrocervical trunk: the inferior thyroid artery, suprascapular artery, and transverse cervical artery. Use color Doppler imaging at least once before inserting the needle to locate any vessel that might be in the path of the needle. Anatomical variations are common.
• Never inject against high resistance because such resistance may indicate needle–nerve contact or an intrafascicular injection. A high opening injection pressure (> 15 psi) is always present with needle–root contact. Thus, a seemingly extraneural injection may, in fact, be subepineural. An intraneural injection may spread proximally into the spinal canal.
• One useful maneuver to ensure injection into the proper compartment, after injecting 5–7 mL, is to trace the plexus down to the supraclavicular fossa (while keeping the needle steady to avoid injury). If the injection is performed inside the brachial plexus “sheath,” the correct spread can be seen very clearly (see Figure 8-A). The probe can then be moved back until the needle is visualized in order to complete the injection. If the brachial plexus appears unchanged in the supraclavicular area, one must question whether the injection was done outside the correct compartment (see Figure 8-B).
• The lateral-to-medial insertion is often chosen to prevent injury to the phrenic nerve, which is typically located anteriorly to the anterior scalene, although one should be aware that the dorsal scapular nerve and the long thoracic nerve usually course through the middle scalene and could potentially be injured as well (Figure 9).
• It is common for C6 and C7 to split proximally. It is prudent to avoid injecting between the nerves coming from a single root, as this may result in an intraneural injection. Instead, it is safer to inject between C5 and C6, superficial to C5, or deep to C6 (Figure 10).
• Another relatively common anatomical variation involves the C5 root traveling through the anterior scalene for part of its course (Figure 11). To nerve block this anatomical variant, the root should be traced distally until it enters the interscalene groove.
• Multiple injections are best avoided as they are not usually needed to successfully nerve block the brachial plexus and may carry higher risk of nerve injury.
• In an adult patient, 7–15 mL of local anesthetic is usually adequate for a successful and rapid onset of block. Smaller volumes of local anesthetic may also be effective;12,13 however, the rate of success of smaller volumes in everyday clinical practice may be inferior to those reported in meticulously conducted clinical trials.
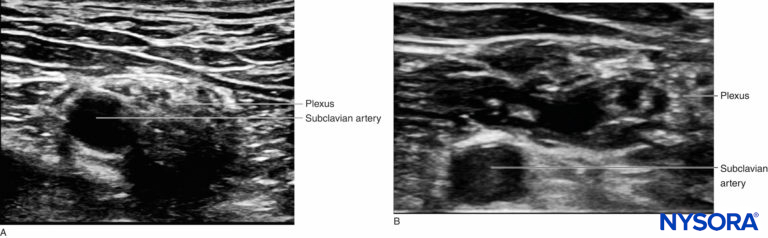
FIGURE 8. Diffusion of local anesthetic solution to the supraclavicular area during interscalene nerve block performance. (A) Before injection. (B) After injection of 10 mL at the interscalene level. The nerves lateral to the subclavian artery are surrounded by local anesthetic and appear deeper. This confirms that the injection was performed in the correct space.
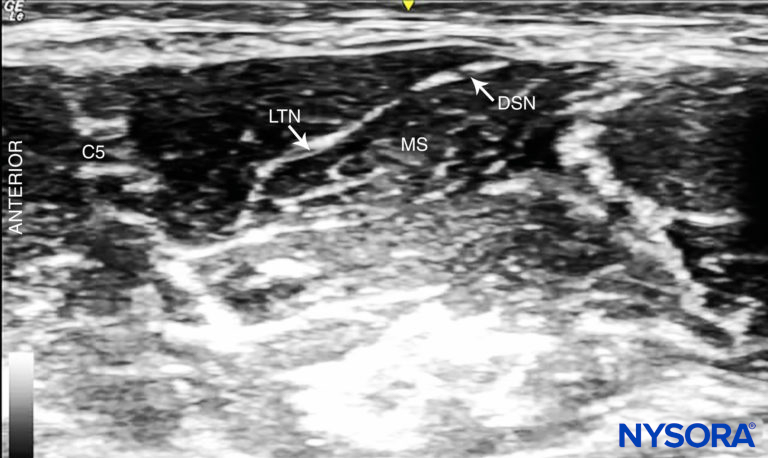
FIGURE 9. Dorsal scapular nerve (DSN) and long thoracic nerve (LTN) visible in the middle scalene muscle (MS).
PHRENIC NERVE block
Phrenic nerve block is common following ISB and may compromise respiratory function in patients with pre-existing pulmonary pathology. Four main strategies14 can be used in such patients to provide analgesia after shoulder surgery while avoiding phrenic block: (1) decreasing local anesthetic volume; (2) performing the ISB more caudad in the neck, around C7; (3) using a supraclavicular nerve block; and (4) using a suprascapular nerve block (possibly in association with an axillary nerve block). The phrenic nerve lies just superficially to the interscalene groove at the level of the cricoid cartilage and courses caudad and anteriorly and distally along the superficial aspect of the anterior scalene muscle along the neck. The use of a low volume of local anesthetic (eg, 5 mL) reduces the incidence of phrenic nerve block but may also reduce the duration of analgesia and possibly decreases the success rate. Higher volumes (10 mL) injected at the level of the cricoid would cause phrenic nerve block.
FIGURE 10. Split C6 and C7 roots in the interscalene groove.
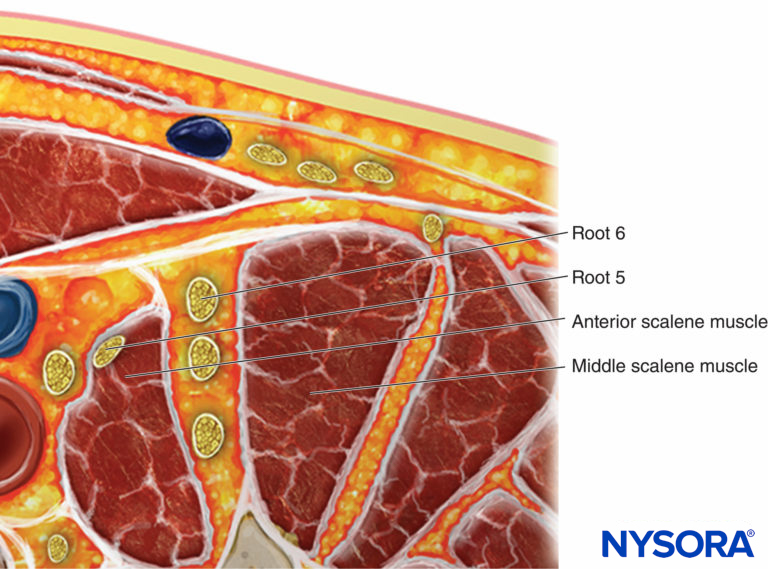
FIGURE 11. Anatomic variation showing the C5 root located in the anterior scalene muscle. To nerve block this anatomical variant, the root should be traced distally until it enters the interscalene groove.
The function of the phrenic nerve can be assessed using ultrasound in M-mode during which a low-frequency curvilinear probe is positioned under the rib cage on the anterior axillary line to evaluate the hemidiaphragm motion (Figure 12). Of note, some authors have recommended using a combination of suprascapular nerve and axillary nerve block to provide postoperative analgesia with minimal motor block distal to the shoulder with a lower risk of phrenic nerve block and other complications of interscalene nerve block. The targeted nerves are small and may prove difficult to find in obese patient. Moreover, these nerve blocks will not provide surgical anesthesia. Another issue that must be considered is persistent phrenic nerve palsy. There is little agreement as to what causes persistent phrenic nerve palsy, but it seems to be at least in part related to inflammation and nerve entrapment rather than direct needle trauma. A contribution of cervical spine disease has been suggested. Other factors may be involved, as most patients in published series have been male, overweight or obese, and middle-aged.
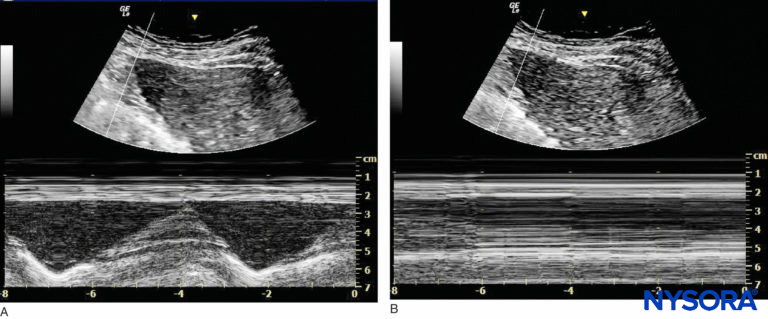
FIGURE 12. Imaging of the right hemidiaphragm under the ribcage on the anterior line. (A) Before interscalene nerve block. (B) After interscalene nerve block with phrenic nerve block.
CONTINUOUS ULTRASOUND-GUIDED INTERSCALENE NERVE BLOCK
The goal of the continuous interscalene nerve block is to place the catheter in the vicinity of the elements of the brachial plexus between the scalene muscles. The procedure consists of: (1) needle placement; (2) LA injection to assure the proper needle tip position and “open up the space” for catheter (3) catheter advancement; (4) Injection through the catheter while monitoring on US to verify its therapeutic position and (5) securing the catheter. For the first two phases of the procedure, ultrasound can be used to ensure accuracy. The needle is typically inserted in plane from the lateral-to-medial direction and underneath the prevertebral fascia to enter the interscalene space (Figure 13), although other needle orientations, such as out-of-plane or aiming caudad, may also be used. Proper needle placement can also be confirmed by obtaining a motor response of the deltoid muscle, arm, or forearm (0.5 mA, 0.1 msec), at which point 4–5 mL of local anesthetic can be injected. This small dose of local anesthetic serves to ensure adequate distribution of local anesthetic as well as to make the advancement of the catheter more comfortable to the patient. This first phase of the procedure does not significantly differ from the single-injection technique.
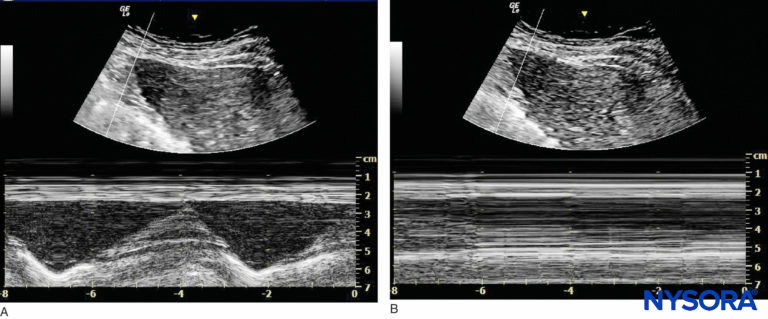
FIGURE 13. Imaging of the right hemidiaphragm under the ribcage on the anterior line. (A) Before interscalene nerve block. (B) After interscalene nerve block with phrenic nerve block.
The second phase of the procedure involves maintaining the needle in the proper position and inserting the catheter 2–3 cm into the interscalene space within the vicinity of the brachial plexus (Figure 14). Catheter insertion can be accomplished by a single operator or with an assistant. Proper catheter location can be confirmed either by visualizing the course of the catheter or by an injection of local anesthetic through the catheter. When this proves difficult, an alternative is to inject a small amount of air (1 mL) to confirm the catheter tip location. The catheter is secured either by taping to the skin with or without tunneling. Some clinicians prefer one over the other. However, the decision about which method to use can be based on patient age, duration of catheter therapy, and/or anatomy. Tunneling may be preferred in older patients with obesity or sagging skin over the neck or when a longer duration of catheter infusion is expected. Two main disadvantages of tunneling are the risk of catheter dislodgement during tunneling and the potential for scar formation. A number of catheter-securing devices are available to help stabilize the catheter. For additional information see Continuous Peripheral Nerve Blocks: Local Anesthetic Solutions and Infusion Strategies.
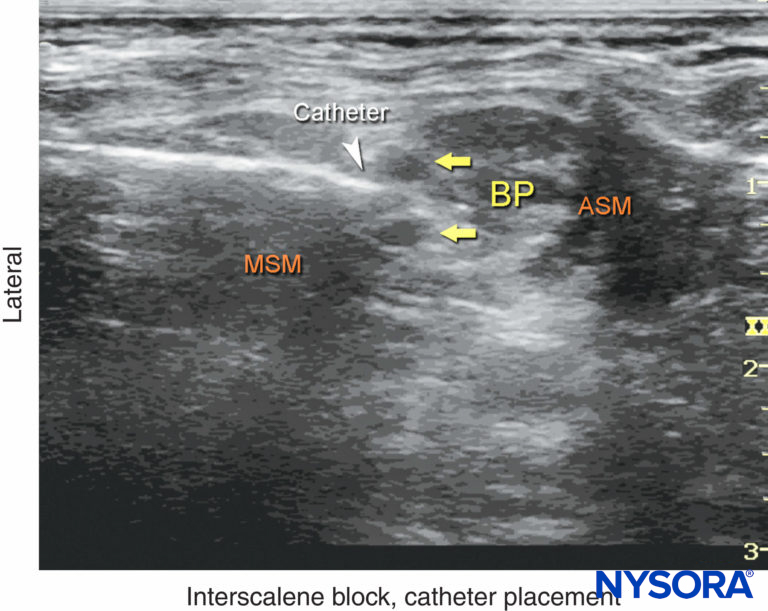
FIGURE 14. An ultrasound image demonstrating needle and catheter (white arrowhead) inserted in the interscalene space between the anterior (ASM) and middle (MSM) scalene muscles. BP, brachial plexus.
TIPS
• Both stimulating and nonstimulating catheters can be used. Because motor response on catheter stimulation may be absent even with ideal catheter placement, the use of stimulating catheters may lead to unnecessary needle and catheter manipulation to obtain evoked motor responses.
• The catheter-over-needle technique has recently been reintroduced as an alternative to accomplishing continuous interscalene block.
To learn more about the Interscalene Nerve Block, see Interscalene Brachial Plexus Block – Landmarks and Nerve Stimulator Technique.
Supplementary video related to this nerve block can be found at Ultrasound-Guided Interscalene Brachial Plexus Nerve Block and Reverse Anatomy Video.
Clinical updates
Kim et al. (Anesthesiology, 2022) report that for ambulatory shoulder surgery, interscalene blocks using perineural liposomal bupivacaine provide analgesia that is noninferior to standard bupivacaine with perineural dexamethasone over the first 72 postoperative hours, with similar block duration (~26–27 h), motor/sensory resolution, and opioid consumption. Although pain scores at some early time points were statistically lower with liposomal bupivacaine, differences did not reach clinical significance, and no meaningful advantages in recovery or adverse events were observed. Given equivalent analgesic efficacy but substantially higher cost, the authors conclude that bupivacaine with dexamethasone can reasonably replace liposomal bupivacaine for routine interscalene analgesia.
Xu et al. (Anesthesia & Analgesia, 2024) describe an anterior glenoid block combined with a selective anterior suprascapular nerve block as a potential phrenic-sparing alternative to interscalene block for shoulder arthroscopy. In 45 patients undergoing arthroscopic rotator cuff repair, 87% reported mild pain (NRS ≤4) within 24 h, while diaphragmatic excursion and handgrip strength were preserved at >80% of baseline in >85% of patients, with no block-related complications. These early data suggest the anterior glenoid block may provide effective analgesia while minimizing diaphragmatic and motor impairment, warranting randomized trials against established shoulder block techniques.