Learning objectives
- Define and describe autonomic dysreflexia
- Recognize signs and symptoms of autonomic dysreflexia
- Manage patients with autonomic dysreflexia
Definition & mechanisms
- Autonomic dysreflexia is a condition that emerges after a spinal cord injury, usually when the damage has occurred above the T6 level
- Dysregulation of the autonomic nervous system leads to an uncoordinated sympathetic response that may result in a potentially life-threatening hypertensive episode when there is a noxious stimulus below the level of the spinal cord injury
- Noxious stimuli consist usually of bladder or bowel distension
- The higher the injury, the greater the severity of the cardiovascular dysfunction
- Significantly increased risk of stroke by 300% to 400%
Signs & symptoms
- Severe headache
- Hypertension
- Piloerection above the level of injury
- Bradycardia
- Facial flushing
- Pallor
- Cold skin
- Sweating in the lower part of the body
- Visual disturbances
- Constricted pupils
- Nasal stuffiness
- Anxiety or feelings of doom
- Nausea and vomiting
- Dizziness
Evaluation
- Identify patients at risk (injury above T6)
- Document baseline blood pressure
- When severe headache occurs, measure blood pressure
- A systolic blood pressure >150 mmHg or >40 mmHg above baseline is indicative of autonomic dysreflexia
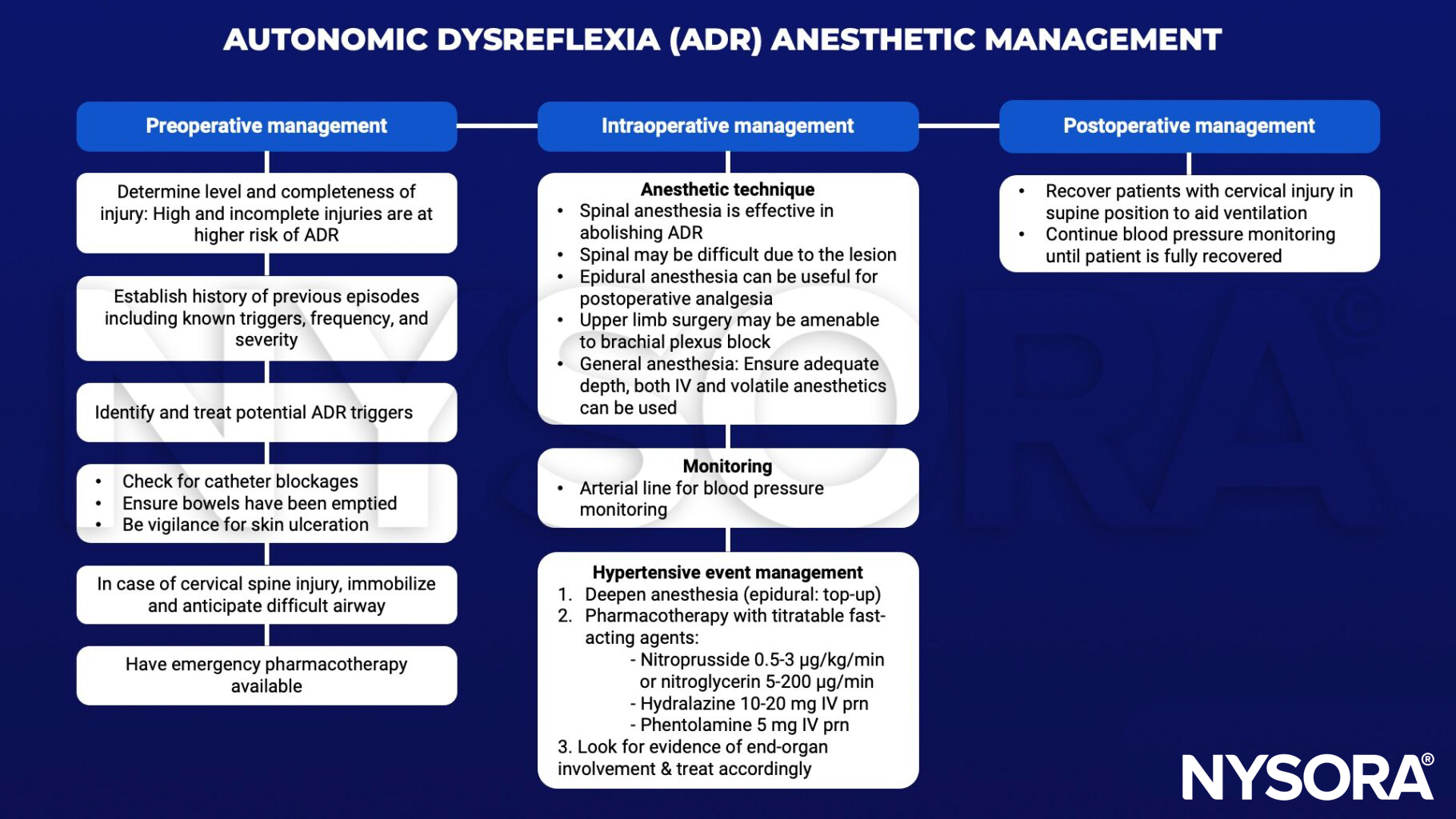
Anesthetic management
Suggested reading
- Allen KJ, Leslie SW. Autonomic Dysreflexia. [Updated 2022 Nov 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482434/
- Petsas A, Drake J. Perioperative management for patients with a chronic spinal cord injury. BJA Education. 2015;15(3):123-30.
Clinical updates
Soriano et al. (Nature, 2025) map the aberrant spinal circuit that emerges after spinal cord injury and underlies autonomic dysreflexia. Following injury, nociceptive sensory neurons establish new connections with excitatory V2a interneurons in the lumbosacral spinal cord, which in turn project to V2a interneurons within the lower thoracic “haemodynamic hotspot.” These thoracic interneurons engage sympathetic preganglionic neurons, creating a maladaptive network capable of generating abrupt, life-threatening hypertensive episodes in response to peripheral stimuli. Targeted epidural electrical stimulation over the lower thoracic haemodynamic hotspot engages this same thoracic V2a population through large-diameter proprioceptive afferents, shifting synaptic balance in their favor. With daily stimulation, aberrant projections from the lumbosacral cord are reduced, beneficial afferent inputs are strengthened, and autonomic dysreflexia is eliminated in mice and rats, while dysreflexia-related symptoms decline in individuals with chronic spinal cord injury. In contrast, stimulation delivered to the lumbosacral cord reinforces pathological circuitry and exacerbates dysreflexia, underscoring the critical importance of precise anatomical targeting for safe and effective neuromodulation.
Trueblood et al. (Neuroscientist, 2024) provide an updated synthesis of the neural and hormonal mechanisms underlying autonomic dysreflexia after high-level spinal cord injury, highlighting how loss of supraspinal control over sympathetic preganglionic neurons, combined with maladaptive sprouting of primary afferents and propriospinal interneurons, amplifies spinal sympathetic reflexes and drives hypertensive crises. The review emphasizes emerging evidence that the renin–angiotensin system (RAS) becomes upregulated after injury and may contribute to baroreflex dysfunction and dysreflexic hypertension, although data remain conflicting regarding which RAS arm is involved. Importantly, the authors outline translational therapeutic strategies, including transplantation of embryonic brainstem- or raphe-derived presympathetic neurons to reestablish serotonergic and noradrenergic regulation of sympathetic output, which reduces spontaneous and induced dysreflexia in rodent models. They propose that combining cell-based restoration of supraspinal control with targeted RAS modulation may offer a mechanism-based approach to rebalance neural and hormonal control of blood pressure after spinal cord injury.
Harmison et al. (Nursing, 2023) provide an updated clinical review of autonomic dysreflexia (AD) in patients with spinal cord injury at or above T6, emphasizing that a systolic BP rise ≥20 mm Hg above baseline signals AD and ≥150 mm Hg or ≥40 mm Hg above baseline constitutes severe AD requiring urgent treatment. They highlight the “six Bs” (bladder, bowel, back passage, boils, bones, babies) as common triggers and outline stepwise management: immediate upright positioning, removal of noxious stimuli, and use of rapid-onset agents such as immediate-release nifedipine or 2% nitroglycerin paste, with BP monitoring every 2-5 minutes until stable. The authors further detail practical nursing interventions, including prompt catheter replacement rather than milking, structured digital stimulation protocols, daily comprehensive skin assessment, and caution with recent PDE5 inhibitor use to prevent recurrence and reduce life-threatening complications.
Hubbard et al. (The Journal of Spinal Cord Medicine, 2021) describe two cases of spinal cord injury patients who developed posterior reversible encephalopathy syndrome (PRES) secondary to autonomic dysreflexia (AD), triggered by urinary tract infections, presenting with seizures, visual changes, and marked blood pressure elevations from low SCI baselines. MRI findings confirmed PRES, and prompt treatment of hypertension with hydralazine plus removal of the AD trigger led to symptom resolution, emphasizing that even “moderate” systolic pressures (e.g., 150–190 mmHg) in SCI patients can overwhelm impaired cerebral autoregulation and precipitate PRES.