INTRODUCTION
Brachial plexus block at the level of the axilla is typically chosen for anesthesia of the distal upper limb. The axillary block is one of the most common approaches to brachial plexus block. Easy landmarks and simplicity make this block suitable for a wide range of surgical procedures.
HISTORY
The surgical technique of this block was first described by William Hallstead in New York City (Roosevelt Hospital, also the clinical affiliation for NYSORA 1995-2014) in 1884, whereas the percutaneous technique was described by Georg Hirschel in 1911. In 1958, Preston Burnham recognized that filling the neurovascular “sheath” with local anesthetic could simplify the axillary block. He also described the characteristic fascial “click” felt upon needle entry into the axillary sheath. In 1961, while using the formula for a cylinder volume, Rudolph De Jong calculated that in an average adult, 42 mL of local anesthetic (LA) was necessary to fill the fascial compartment to the level of the cords and block all terminal nerves to the arm. A year later, Ejnar Eriksson and Skarby, in an effort to promote the proximal spread of LA, advocated wrapping a rubber tourniquet around the arm, distal to the needle.
In 1979, Alon Winnie and coworkers found the tourniquet ineffective and painful and recommended firm distal digital pressure on the neurovascular sheath instead. In addition, they also recommended arm adduction after LA injection, thinking that the head of the abducted humerus compressed the neurovascular sheath. Both maneuvers were later proved to be clinically ineffective. Gale Thompson and Duane Rorie, in 1983, studied brachial plexus using computed tomograms and suggested that the median, ulnar, and radial nerves lie in separate fascial compartments within the neurovascular sheath; this hypothesis provided a rational explanation for incomplete blocks. However, anatomic studies by Lassale and Ang in 1984 and Vester-Andersen and coworkers in 1986 did not confirm the existence of a true neurovascular sheath. The interfascial space they found contained the median and the ulnar nerves, infrequently the musculocutaneous, and occasionally the radial nerves. Moreover, the space was suggested to communicate proximally only with the medial cord of the plexus. In 1987, Partridge and coworkers found the interneural septa, which were easily broken by injection of dyed latex.
In 2002, Oivind Klaastad and coworkers were the first to investigate the spread of the LA through the axillary catheter in studies using magnetic resonance imaging (MRI) scanning. They found that in most patients the spread of LA was uneven and the clinical effect inadequate. Until the 1960s, the prevalent block techniques were double or multiple axillary injections. After the concept of the neurovascular sheath had been established by De Jong in 1961, the single-injection technique, being the simplest, became standard. However, Vester-Andersen and coworkers demonstrated in 1983 and 1984 that despite high volumes of LA, analgesia was often inconsistent (“patchy”). In the early 1990s, the double-injection, transarterial technique was popularized by Urban and Urquhart17 and Stan and coworkers. More recently, however, the development of peripheral nerve stimulators and insulated atraumatic needles has allowed electrolocation and separate block (multi stimulation technique) of the individual terminal nerves (median, musculocutaneous, ulnar, and radial). This is known as the multiple-nerve stimulation technique. Baranowski and Pither (in 1990), Lavoie and coworkers20 (in 1992), Koscielniak-Nielsen and coworkers (in 1997 and 1998), and Sia and coworkers (in 2001 and 2002) independently showed that multiple-nerve stimulation was superior, both to the single- and the double-injection methods by increasing the success rate and shortening the block onset. A recent Cochrane review by Handoll and coworkers validated these findings.
INDICATIONS AND CONTRAINDICATIONS
The most common indications for axillary block include surgery of the forearm, wrist, or hand of moderate to long duration, with or without an arm tourniquet. Relative contraindications to the use of this block are skin infection at the block site, axillary lymphadenopathy, and severe coagulopathy. In addition, this block is best avoided in patients with preexisting neurologic disease of the upper extremity because sensory assessments may be difficult.
PERTINENT ANATOMY
In the apex of the axilla, the three plexus cords (lateral, medial, and posterior) form the main terminal nerves of the upper extremity (axillary, musculocutaneous, median, ulnar, and radial). However, only the last three nerves accompany the blood vessels through the axilla where the blocks are performed (Figure 1), while the axillary and the musculocutaneous nerves leave the plexus approximately at the level of the coracoid process. The axillary nerve departs at a wider angle from the posterior cord, laterally and dorsally, and the musculocutaneous nerve, which originates from the lateral cord, runs obliquely laterally into the coracobrachialis muscle and continues downward. The medial antebrachial cutaneous and brachial cutaneous nerves run subcutaneously parallel to the axillary vessels, although the medial antebrachial cutaneous nerve often follows the median nerve within the neurovascular sheath. In the axilla, the median and musculocutaneous nerves lie superior to the artery, whereas the ulnar and radial nerves lie inferior to it.
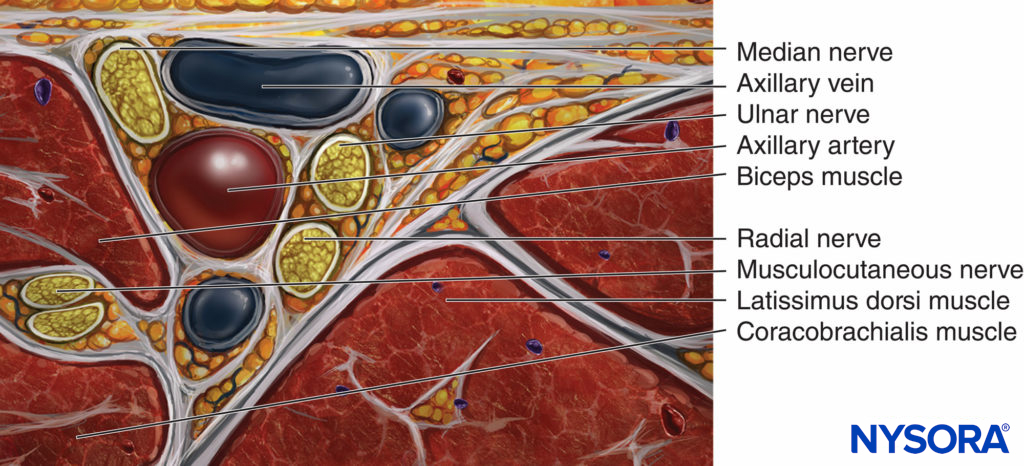
FIGURE 1. Anatomy of the brachial plexus at the axilla and at the mid humeral level.
The depths at which the nerves are found vary. Typically, the median nerve is more superficial than the musculocutaneous, and the ulnar nerve is more superficial than the radial. Occasionally, the radial or the musculocutaneous nerves (or both) are found behind the artery. These two nerves progressively diverge from the neurovascular sheath, continuing down the upper arm, the musculocutaneous above (anterior) and the radial below (posterior) to the humerus, where they can be approached using the mid-humeral approach.
Landmarks
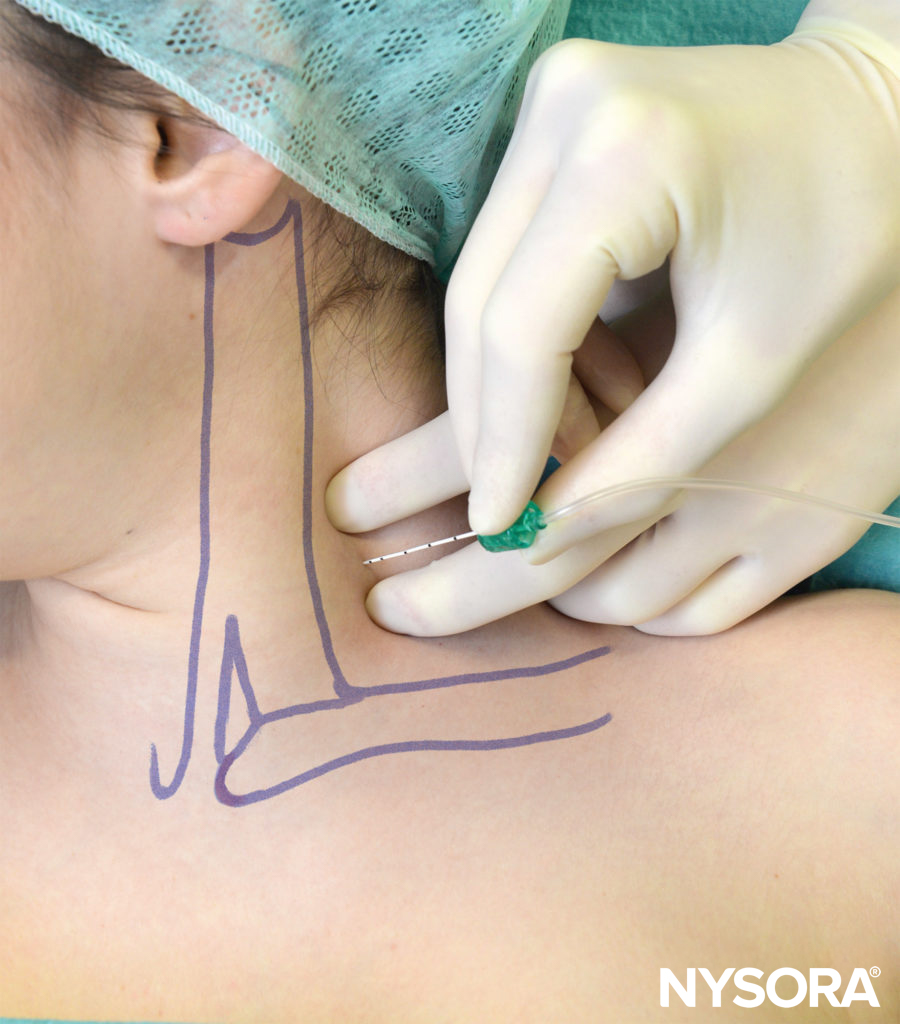
Surface landmarks for the axillary brachial plexus block include (Figure 2):
- The pulse of the axillary artery
- Coracobrachialis muscle
- Pectoralis major muscle
- Biceps muscle
- Triceps muscle
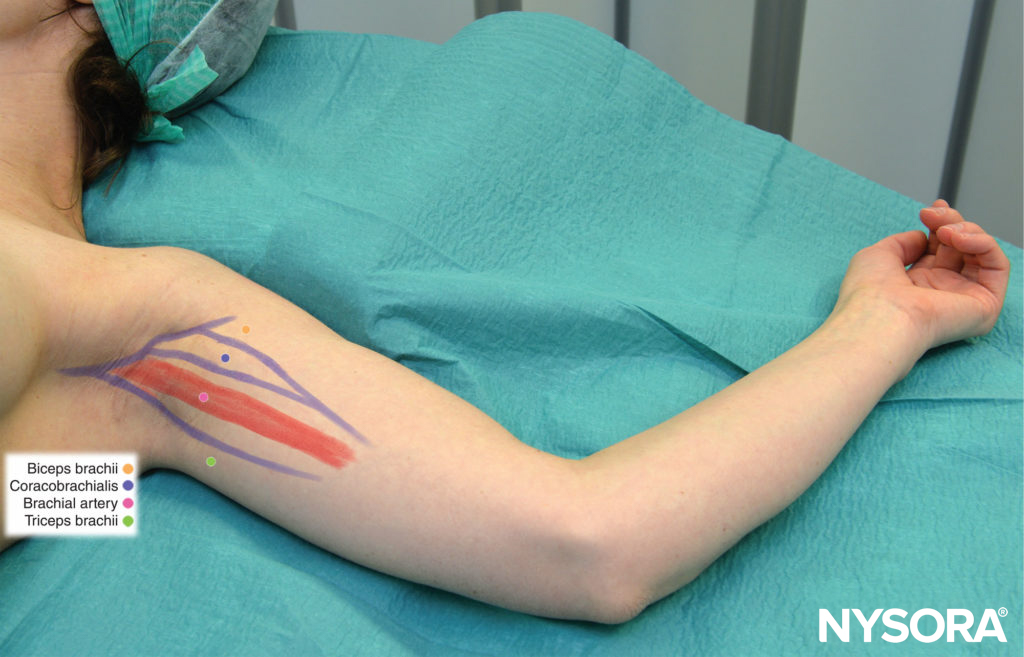
FIGURE 2. Landmarks for the axillary brachial plexus block.
EQUIPMENT
- Sterile towels and 4-in. × 4-in. gauze packs
- Sterile gloves, marking pen, and a skin electrode
- 1-in., 25-gauge needle for skin infiltration
- 1- to 1.5-in. atraumatic, insulated stimulating needle
- 20-mL syringes containing LA of choice
- Peripheral nerve stimulator
- Means of assessing opening injection pressure
Learn more about Equipment for Regional Anesthesia.
INJECTION TECHNIQUES
Arm Position for the Block
The arm to be operated on is abducted approximately 90 degrees (see Figure 2). The elbow is flexed and the forearm rests comfortably, supported by a pillow. The arterial pulse is palpated at the level of the major pectoral muscle, and the subcutaneous tissue overlying the artery is infiltrated with 4–5 mL of LA (to block the intercostobrachial and medial cutaneous nerves of the arm). Several techniques and approaches to the brachial plexus block at the level about the axilla have been described; we will describe only some of the well-studied techniques. A triple-injection axillary block is probably the most efficient technique for axillary brachial plexus block.
NERVE STIMULATION TECHNIQUES
Single-Injection (Stimulation) Technique
- The nerve stimulator is set to deliver 0.5–1.0 mA (2 Hz, 0.1 msec); electrical connections with the needle and the neutral electrode are checked.
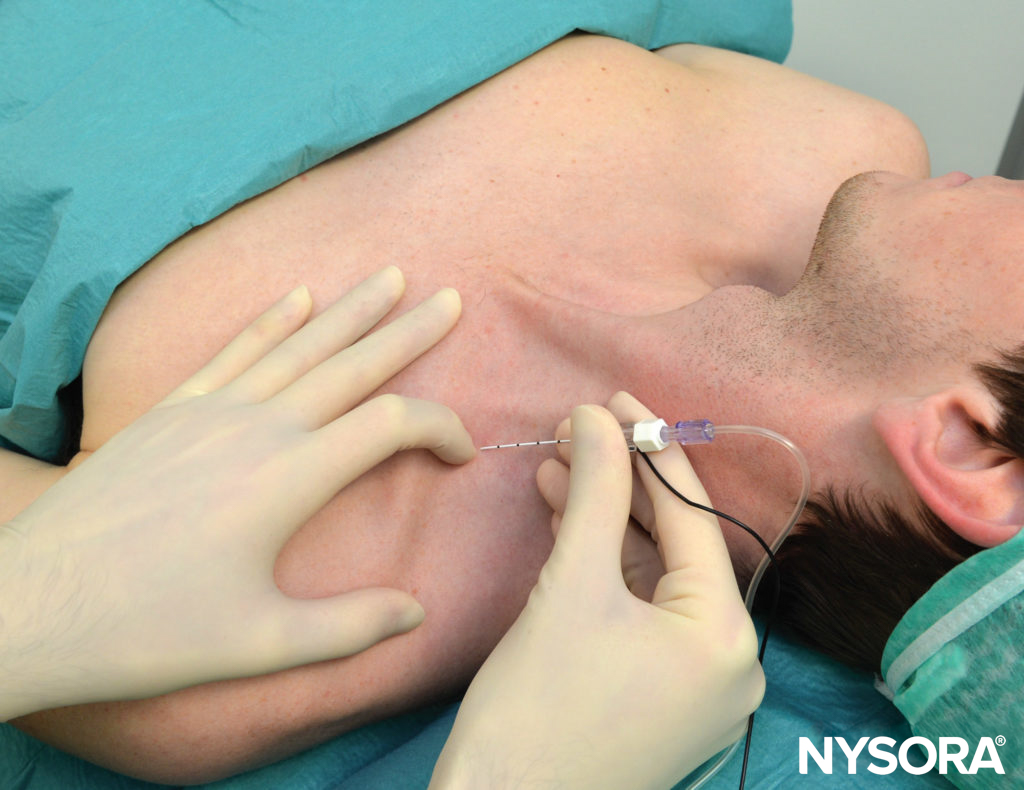
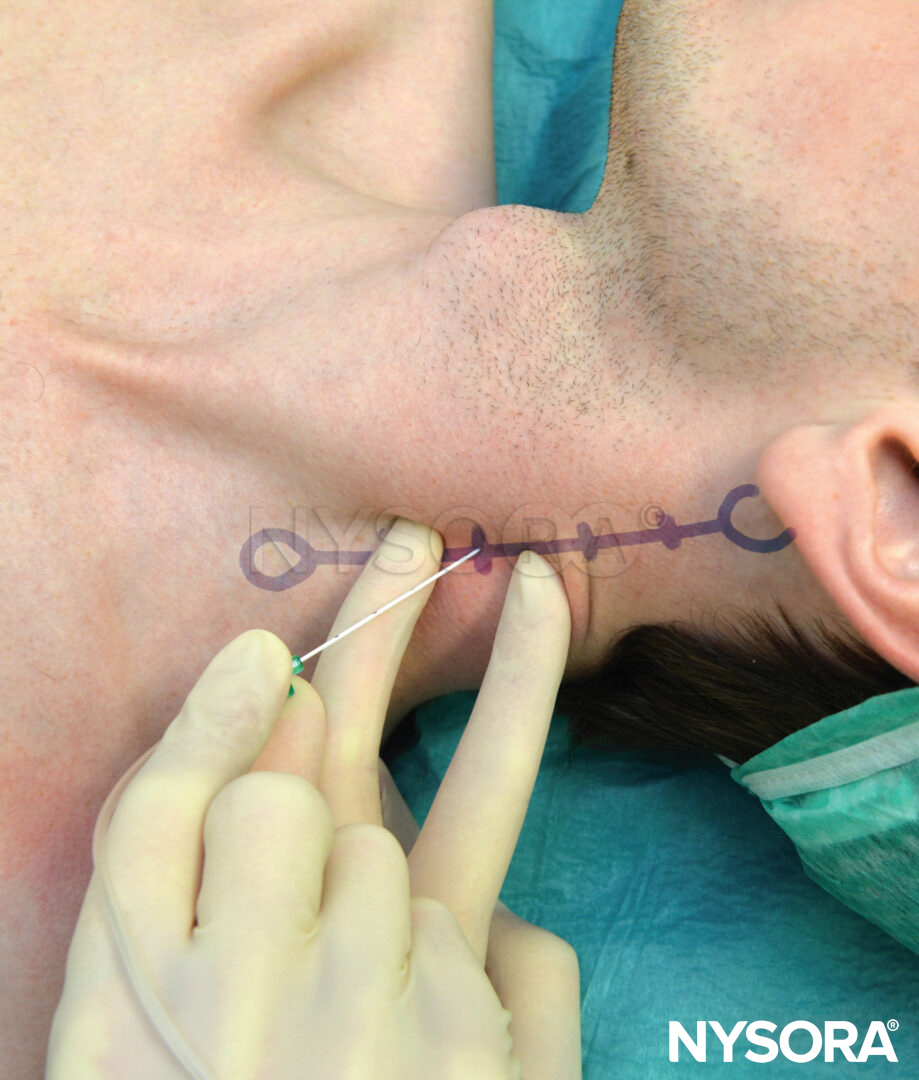
- Depending on the surgical site (palmar and medial or dorsal and lateral aspects of the hand/forearm), the stimulating needle is inserted above the arterial pulse (toward the median nerve) or below the arterial pulse (toward the radial nerve), respectively (Figure 3).
- As the superficial fascia is penetrated, a characteristic “click” is often felt, and the current amplitude is slowly increased (e.g., at 1-mA increments) until the desired twitch (flexion or extension of the wrist and fingers) is obtained. This helps avoid painful electrical paresthesia when the elastic fascia suddenly “gives in” and the needle enters the neurovascular sheath.
- After the initial motor response is obtained, the needle is slowly advanced toward the stimulated nerve while reducing the amplitude.
- Once the stimulation is obtained using a current intensity of 0.3–0.5 mA, the entire volume of LA is injected slowly, while intermittently aspirating to reduce the risk of accidental intravascular injection. This results in a substantial spread of the LA within the tissue layers encompassing the brachial plexus (Figure 5).
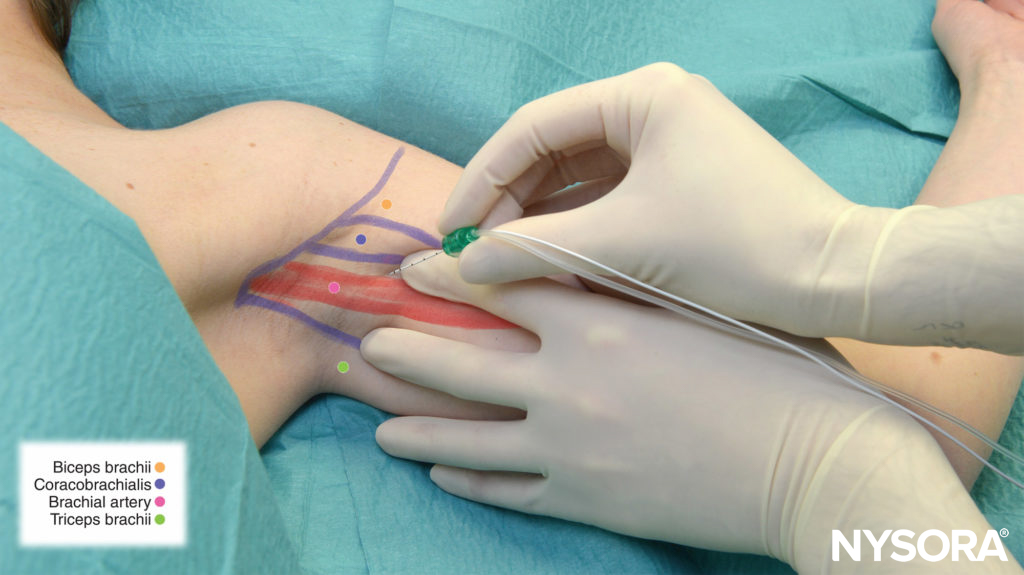
FIGURE 3. Median nerve block: The needle is inserted above the pulse of the axillary (brachial) artery.
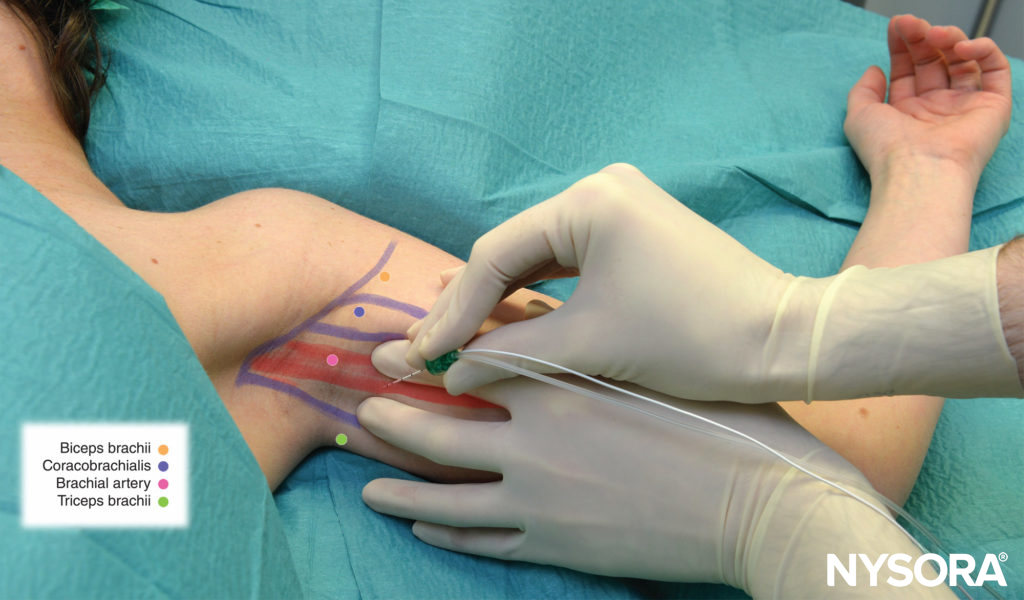
FIGURE 4. Radial nerve block: The needle is inserted below the pulse of the axillary (brachial) artery.
NYSORA Tips
- Arterial pulse palpation may prove challenging in some patients. In these patients, the initial motor response can be used to guide needle redirection to achieve the desired response.
- Elbow flexion (stimulation of the coracobrachialis muscle or the musculocutaneous nerve) indicates that the needle is outside the neurovascular sheath; the needle should be redirected downward and more superficially.
- Extension of the wrist and hand (radial nerve) indicates that the needle is below the artery; the median and ulnar nerves are above the artery.
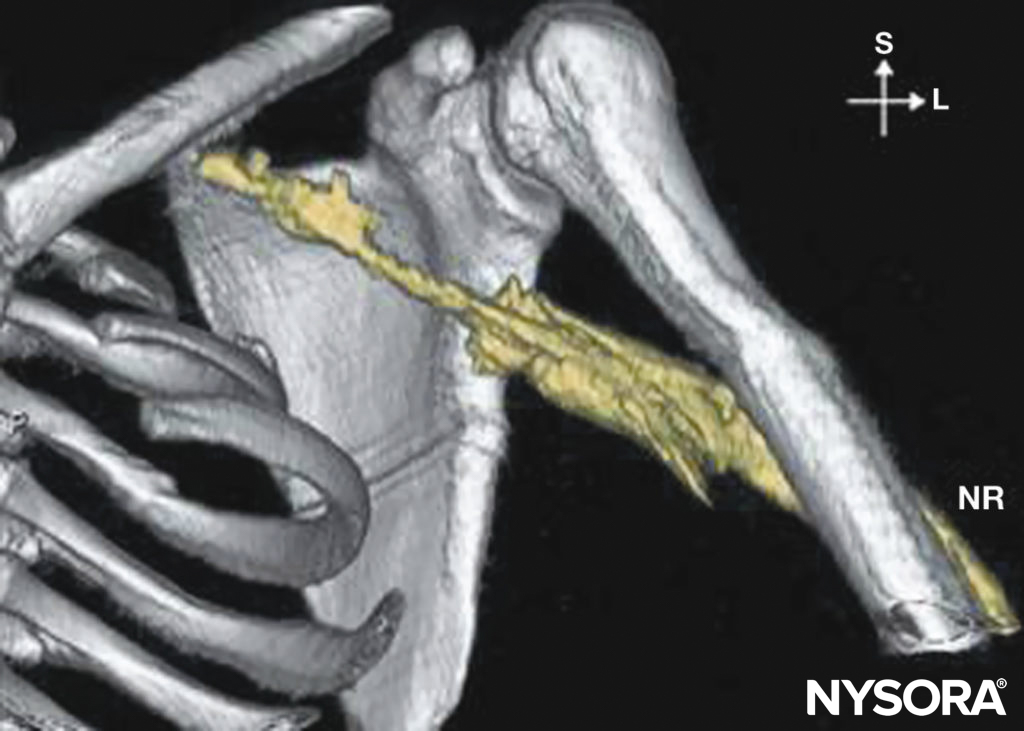
FIGURE 5. Distribution of local anesthetic injected during axillary brachial plexus block. NR = nervus radialis.
- The more difficult differentiation is between the median and the ulnar nerves, which both result in wrist/finger flexion. In this scenario, the following method can be used to differentiate between the two nerves:
- When flexion is accompanied by forearm pronation, the stimulated nerve is the median (the needle is positioned above the artery).
- Another way to differentiate between these two nerves is by palpation of the flexor tendons at the wrist. Median nerve stimulation produces movements of the palmaris longus and the flexor carpi radialis tendons, which lie in the middle of the wrist, whereas ulnar nerve stimulation produces movement of the flexor carpi ulnaris tendon, which lies medially.
- Decreasing the intensity of the output current of the nerve stimulator helps facilitate differentiation between the median and ulnar nerve stimulation.
Double-Injection Technique
- The stimulating needle is first inserted above the artery, below the coracobrachialis muscle (see Figure 3). After penetrating the fascia, the amplitude is increased until synchronous wrist flexion/pronation and flexion of the first three fingers are obtained (median nerve stimulation). The needle is advanced slowly toward this nerve while reducing the amplitude to 0.3 to 0.5 mA. At this point, half of the planned volume of LA is slowly injected with an intermittent aspiration to rule out an intravascular injection.
- The needle is then withdrawn and inserted below the artery and above the triceps muscle (see Figure 4). The fascia is again penetrated and the amplitude slowly increased. The first response is usually either arm extension (muscular branches to the triceps) or thumb adduction and flexion of the last two fingers (ulnar nerve). However, these responses are ignored, and the needle is advanced deeper, often slightly upward, behind the artery (Figure 6) until the wrist and finger extension is obtained (radial nerve). After stimulation is obtained using a current intensity lower than 0.5 mA, the remaining volume of LA is slowly injected with intermittent aspiration.
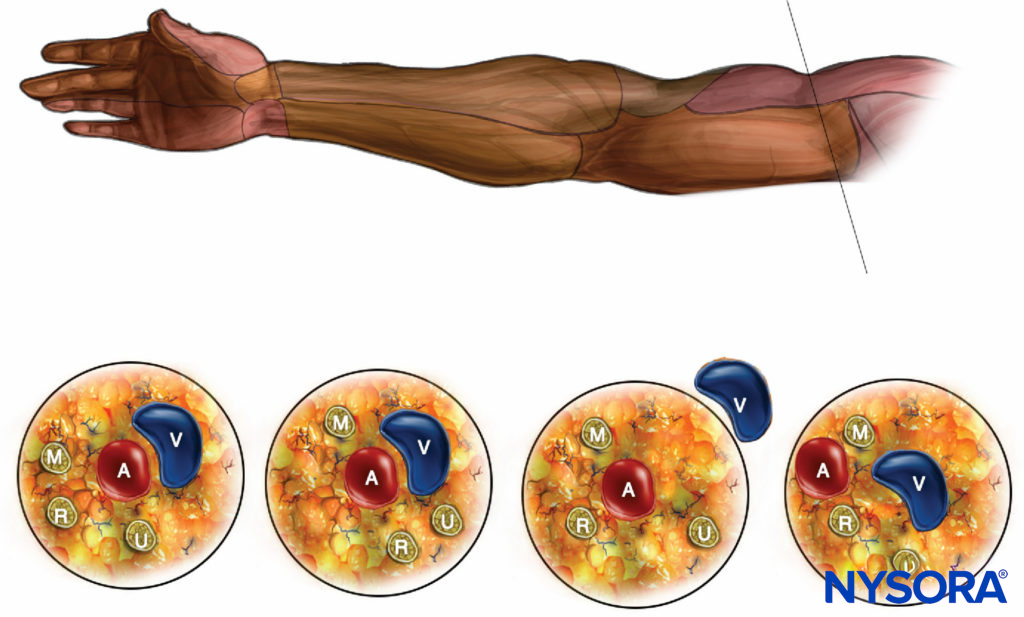
FIGURE 6. The spatial arrangement of the terminal nerves of the brachial plexus in the axilla. M = median nerve, A = artery, V = vein, R = radial nerve, U = ulnar nerve.
NYSORA Tips
- With ultrasound-guided nerve blocks, motor stimulation below 0.5 mA is avoided to decrease the risk of a needle–nerve contact or intraneural injection. With nerve stimulator–guided blocks, however, a motor response is sought at 0.3—0.5 mA as evoked motor response is the only means of nerve localization, and nerve stimulation does not allow visualization of the tissue spaces or spread of the local anesthetic which is possible with ultrasound guidance.
Multiple-Injection Technique
Needle insertion sites are identical to those for the double injection technique.
- After electrolocation of the median nerve, 5–10 mL of the LA volume is injected (see Figure 3).
- The needle is withdrawn subcutaneously and redirected obliquely, above and into the coracobrachialis muscle. After obtaining stimulation-synchronous biceps flexion, the amplitude is reduced to 0.3–0.5 mA and another 5–10 mL of LA is injected to block the musculocutaneous nerve.
- The needle is removed and inserted below the artery (see Figure 4). The first stimulated nerve is usually the ulnar nerve, into which 5–10 mL of LA is injected.
- The needle is advanced deeper until the radial nerve is found.
NYSORA Tips
- Two studies by Sia and colleagues suggest that two separate injections below the artery do not improve success rates, and therefore only one such injection is needed. This injection is made close to the radial nerve and should contain half of the planned LA volume.
- Electrolocation of multiple nerves may occasionally take some time. Because the first injection of the LA injection in the vicinity of the median nerve may partially block the ulnar nerve, the search for the nerves should be made expeditiously to minimize the risk of needle nerve contact or intraneural injection into an anesthetized nerve.
- For these reasons, this technique could be considered an advanced regional anesthesia technique. Careful assessment of resistance to injection by an experienced practitioner or objective monitoring of injection pressure should be used with each injection.
Transarterial Technique
- This relatively simple technique does not rely on a nerve stimulator; instead, placement of the needle within the neurovascular sheath is identified by relying on the axillary artery:
The axillary artery is palpated and stabilized using a two-finger palpation technique. - As the needle is advanced toward the pulse of the axillary artery, bright red arterial blood is aspirated. A thin, long beveled needle (typically 1.5-in., 25-gauge) is used to minimize the risk of axillary hematoma.
- The needle is advanced deeper until blood cannot be aspirated (the tip of the needle has exited the artery) and half of the volume of the LA is injected behind the posterior wall. This should block the radial nerve.
- The needle is slowly withdrawn while aspirating. As the needle enters the axillary artery, bright red blood is again aspirated.
- The withdrawal of the needle is continued until blood cannot be aspirated (the needle exits the artery and its tip is positioned superficial [media] to the artery inside the neurovascular sheath).
- The remaining volume of LA is injected superficial to the anterior wall to block the median and the ulnar nerves.
- A transarterial injection is made as high up in the axilla as possible, and the needle should traverse the artery at an oblique angle. This reduces the risk of making the injection behind the artery intramuscularly and improves the spread of the LA to the plexus cords to block the musculocutaneous nerve.
MIDHUMERAL APPROACH (HUMERAL CANAL BLOCK)
The difference between the multi injection axillary and the mid humeral (humeral canal) approaches is that in the latter, two-terminal nerves, the musculocutaneous and the radial, are blocked separately, above and below the humeral bone, respectively (Figures 1 and 7). With any multi stimulation technique, there is always a risk that an intraneural injection may be made into the already anesthetized nerves. Although the four-injection mid humeral block has been found to be more effective than the double-injection axillary technique, either block results in a very high success rate when four injection techniques are used. An advantage of the axillary approach is that incomplete axillary blocks can be supplemented with a mid humeral block. The opposite is not possible, nor is it recommended because electrostimulation may be precluded by block distal to the site of nerve localization. An incomplete mid humeral block, on the other hand, can be supplemented at the elbow or the wrist.

FIGURE 7. Spatial arrangement of the terminal nerves of the brachial plexus at the mid humerus.
Technique
The injection technique for the mid humeral block is similar to the four-injection axillary technique, except the injections are made more distally. In addition, the musculocutaneous and the radial nerves are sought at a deeper location than in the axillary approach (see Figure 7). Figure 8 demonstrates the spread of the injected local anesthetic in the mid humeral technique.
- The nondominant hand grips the biceps muscle while searching for the musculocutaneous nerve, and the stimulating needle is inserted below the muscle
(to avoid direct stimulation). - When the bone is contacted before eliciting the twitches, the needle is redirected upward, toward the belly of the biceps muscle.
- The triceps muscle is stabilized similarly while attempting stimulation of the radial nerve. It should be kept in mind that the radial nerve winds around the humeral shaft on its way downward, which makes electrolocation of this nerve challenging with distal approaches.
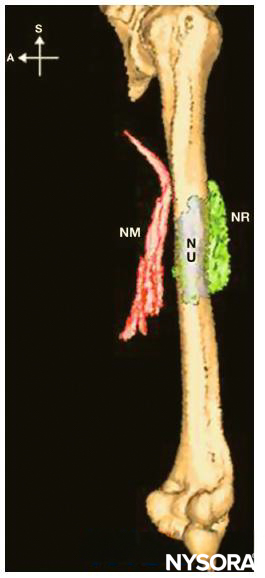
FIGURE 8. Distribution of injectates after midhumeral block. NM = nervus medialis, NU = nervus ulnaris, NR = nervus radialis.
CHOICE OF LOCAL ANESTHETIC
The choice of LA depends on the length of surgery and the desired density and duration of block. For single-injection blocks, short- and medium-acting LAs (prilocaine, 2-chloroprocaine, lidocaine, or mepivacaine) in concentrations of 1.5%–2% (3% for 2-chloroprocaine), with or without epinephrine or sodium bicarbonate, will provide reliable sensory and motor block of rapid onset (10–20 min) and sufficient duration (3–4 h; 1.5–2 h for 2-chloroprocaine) for most acute and subacute procedures (e.g., wound debridement; closed fracture repositions; ligament-, tendon-, or nerve sutures; finger amputations). For elective procedures of longer duration (e.g., arthrodeses, arthroplasties, osteosyntheses, extensive palmar fasciectomies) ropivacaine 0.5%–0.75% or bupivacaine 0.375%–0.5%, with or without epinephrine, will provide analgesia of slightly slower onset (15–20 min) and longer duration (6–16 h). For specialized hand surgery that may last several hours—for example, multiple joint replacements or reimplantations of severed extremities—a continuous ropivacaine (0.2%–0.375%) infusion via an axillary catheter is probably the best technique. Clonidine (0.5 mcg/kg) may be added to intermediate-acting LAs to prolong analgesia after single-shot blocks.
PERIOPERATIVE MANAGEMENT
The multiple-nerve stimulation technique is uncomfortable for patients and should be preceded by adequate premedication (e.g., midazolam + sufentanil). Adequate sedation and analgesia not only improve patients’ acceptance of the block but also help relax the arm muscles. This makes precise needle manipulation as well as eliciting and interpreting the motor responses to nerve stimulation significantly easier for the practitioner and more acceptable to patients.
NYSORA Tips
- The first sign of a successful block is weakness of the upper arm muscles, which can be tested immediately after needle withdrawal. This can be done by asking the patient to place the hand on the abdomen or touch the practitioner’s finger.
- Loss of coordination signifies that the mantle fascicles of the musculocutaneous and radial nerves, which supply flexors and extensors, are being blocked. Very often patients report an early loss of the position sense in the blocked extremity.
The onset and distribution of analgesia can be tested every 5 or 10 minutes after block administration in the sensory areas of the seven terminal nerves (Figure 9). Thirty minutes after block insertion, the unblocked nerves can be supplemented distal to the initial block site (e.g., elbow blocks).
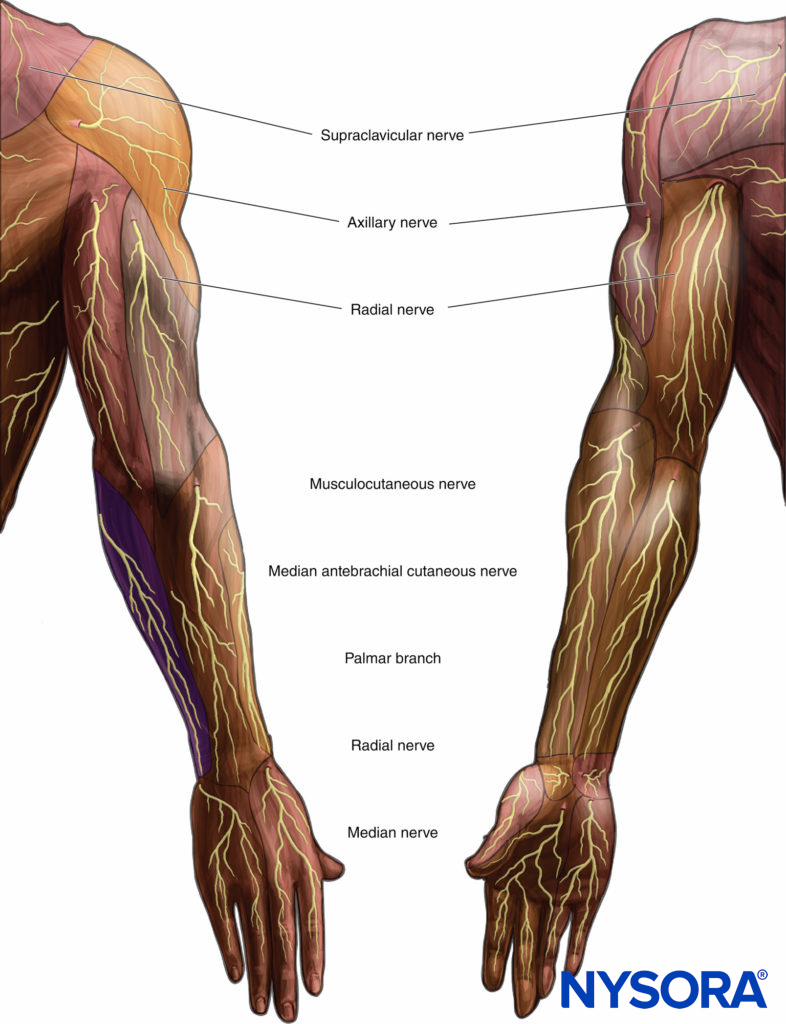
FIGURE 9. Sensory innervation of the upper extremity.
NYSORA Tips
- Most hand surgery (e.g., palmar fasciectomies and nerve or tendon repair) is performed on the volar aspect and theoretically can be performed with partial blocks (i.e., without the radial or musculocutaneous nerves).
- For elbow surgery, an infraclavicular approach is a better choice then the axillary block.
- Tourniquet analgesia may be more related to the total injected dose of LA rather than to successful block of the medial cutaneous brachial nerves. Most of the injected LA is absorbed into the surrounding muscles, which are the main source of ischemic pain.
CONTINUOUS AXILLARY BLOCK
The indications for continuous axillary block include control of acute postoperative pain, management of chronic pain, and treatment of vascular disease, (e.g., Raynaud syndrome).
Technique
The axillary fossa is shaved and disinfected. After subcutaneous LA infiltration, the specific muscle twitch from the nerve of greatest interest is elicited by a needle or stimulating introducer cannula. The intensity of the stimulating current is progressively reduced to 0.5 mA or less, while making fine adjustments to the needle position. A catheter is inserted (under sterile conditions) 5–8 cm cephalad into the neurovascular sheath and either sutured to the skin or tunneled. This helps maintain the catheter in place because the nerves are superficial and the arm sweat makes maintenance of an occlusive dressing difficult.
NYSORA Tips
- Difficulty with catheter insertion usually indicates placement of the needle outside the neurovascular sheath.
Maintenance
Diluted solutions of long-acting LAs (eg, 0.125% bupivacaine or 0.2% ropivacaine) are most often used for continuous infusions. A common infusion regimen includes intermittent bolus of LA (5-10 mL Q 4-6 hrs) of the dilute mixture of LA with or without continuous infusion of 5 mL/h.
NYSORA tips
- A typical infusion regimen for 0.2% ropivacaine is a basal rate of, for example, 0.1 mL/kg bodyweight per hour (minimum, 5 mL; maximum, 10 mL) and a 5 mL patientcontrolled bolus with a lock-out time of 30 minutes.
Complications
Vascular Puncture Vascular puncture can occur with an axillary block but it usually can be detected. However, a venous puncture may be undetected if aspiration or palpation pressure collapses the venous lumen.
Intravascular LA Injection Intravascular LA injection manifests itself as lightheadedness and or tachycardia (ropivacaine- or epinephrine-containing solutions). Note that intra-arterial injection produces hand paresthesia during injection accompanied by sudden paleness. Slow injection with repeated needle aspirations is mandatory.
Hematoma may occur after an arterial puncture. If the artery is punctured, firm, steady pressure should be applied over the puncture site for 5–10 minutes. For the transarterial technique, needles of smaller gauge should be used to minimize the risk of hematoma.
Toxicity Due to Absorption of LA Toxicity due to absorption of LA (in contrast to the accidental intravascular injection, which becomes symptomatic during or immediately after the injection) usually becomes symptomatic 5–20 minutes after injection. The symptoms include lightheadedness, dizziness, tunnel vision, circumoral paresthesia, bradycardia or tachycardia, anxiousness (eventually progressing to unconsciousness), and seizures. Oxygen, a sedative/hypnotic in titrated doses, and airway support if necessary should be immediately administered.
Nerve Injury Nerve injury may be caused by the advancing needle, intraneural injection, application of a tourniquet, or a combination of these. Intraneural injections are characterized by pain, extremity withdrawal, and resistance to injection. Needle and injection injuries typically manifest as neurologic deficits in the distribution of the affected nerve. However, ischemic damage caused by prolonged application of the tourniquet more commonly results in a diffuse injury, affects several nerves, and is usually accompanied by soreness of the upper arm. Symptoms of nerve damage (sensory loss and persistent paresthesia) usually appear within a day or two after recovery from the block. Most nerve injuries are neurapraxia (functional damage), which carry a good prognosis and heal within a few weeks.
NYSORA Tips
- When the motor response to nerve stimulation is seen with currents <0.2 mA, the tip of the needle should be slightly withdrawn or repositioned to maintain the twitch with 02–0.5 mA.
- LA should never be injected when abnormal resistance (high opening pressure) to injection is encountered. When this occurs, the needle should be pulled back slightly and the injection reattempted. If the resistance persists, the needle should be completely withdrawn and cleared; it should never be assumed that the cause of the resistance is related only to needle obstruction.
SUMMARY
For axillary brachial plexus block, a triple-injection nerve stimulator technique with electrolocation of median, musculocutaneous, and radial nerves is preferred. A double- injection technique is the next best and may be used with or without a nerve stimulator. The mid humeral (a four-injection technique) is probably best suited for supplementing incomplete axillary blocks, although it can be used as a primary technique. For continuous blocks, the catheter should be placed close to the main nerve innervating the surgical site (e.g., the median nerve for surgery of medial and volar surfaces; the radial nerve for surgery of lateral and dorsal surfaces). For more extensive surgery involving the entire circumference of the arm (e.g., major trauma/amputation), an approach higher in the axilla or infraclavicular block may be better suited. An optimal perineural infusion technique is a basal infusion plus patient-controlled boluses; the suggested LA for this application is ropivacaine 0.2%. An accidental intravascular injection is the most common complication of an axillary block. The risk of systemic toxicity of LA can be decreased by avoiding fast, forceful injection and using frequent aspiration to rule out an intravascular injection. Pain, paresthesia, extremity withdrawal, or high injection pressure may indicate intraneural needle placement; the occurrence of any of these signs and symptoms should prompt immediate cessation of the injection and reevaluation.
Clinical updates
Nijs et al. (Journal of Clinical Medicine, 2024) report in a systematic review and meta-analysis of 28 RCTs (n≈6,100) that ultrasound-guided axillary brachial plexus block achieves similar rates of adequate surgical anesthesia at 30 minutes compared with supraclavicular block, but a slightly lower success rate than infraclavicular block, with no differences in conversion to general anesthesia or need for supplemental analgesia. Infraclavicular blocks were performed more quickly, whereas onset times were comparable across approaches. Importantly, the axillary brachial plexus block showed a more favorable safety profile, with significantly less Horner’s syndrome and avoidance of pneumothorax or phrenic nerve involvement, supporting the axillary brachial plexus block as a preferred option in patients at higher risk from proximal block–related complications.
- Read more about the study HERE.
Grape et al. (European Journal of Anaesthesiology, 2021) report in a single-blinded randomized trial of 50 patients that an ultrasound-guided single-injection axillary brachial plexus block performed with extreme arm abduction significantly reduces procedure time (≈4 vs 6 min) compared with a multiple-injection technique, while achieving a similar block success rate at 30 minutes. The trade-off was a longer onset time with the single-injection approach, although all failures were readily resolved with supplemental blocks. These findings suggest that a single-injection technique may improve efficiency and reduce needle passes, at the cost of slower onset, and merit further study with block success as a primary outcome.