Rib fractures remain a major source of morbidity in trauma patients, often leading to respiratory compromise, prolonged ICU admission, and increased risk of pneumonia. Effective analgesia is essential: inadequate pain control limits deep breathing, coughing, and mobilization, while opioid-heavy regimens may worsen delirium, hypoventilation, and gastrointestinal dysfunction. These challenges have driven widespread adoption of the erector spinae plane (ESP) block as a lower-risk alternative to epidural and paravertebral approaches. ESP catheters are particularly attractive because they can offer continuous, paravertebral-like analgesia while avoiding neuraxial contraindications common in trauma populations.
However, the success of continuous ESP analgesia depends entirely on the catheter remaining within the correct fascial plane. Although ultrasound confirms initial placement, the true position of the catheter over subsequent days—especially in a population receiving frequent repositioning, chest physiotherapy, and intensive care handling—has been unknown. Unlike peripheral nerve catheters, where migration patterns are well documented, postplacement behavior of ESP catheters has never been systematically evaluated.
This retrospective cohort study examined chest CT scans that were incidentally performed within 120 hours of ESP catheter placement for rib-fracture analgesia. The goal was straightforward yet clinically important: determine whether indwelling over-the-needle ESP catheters actually remain in the plane where they are placed, and, if not, how far they migrate from the intended target.
Study objective and methods
The primary objective was to determine how often indwelling erector spinae plane (ESP) catheters remained correctly positioned within the ESP fascial plane when evaluated on chest CT after placement for rib fracture analgesia.
- Design: Single-center, retrospective cohort study.
- Setting: Surgical ICU, Henry Ford Health Main Hospital (USA).
- Population: Adults admitted with traumatic rib fractures (January 2020–July 2022) who received continuous ESP analgesia via an over-the-needle catheter and underwent at least one chest CT within 120 hours of catheter placement.
- ESP catheter procedure: Ultrasound-guided longitudinal insertion at the targeted thoracic level using an over-the-needle system (E-cath II Plus, Pajunk). Catheters were positioned between the erector spinae muscle and transverse processes, confirmed with hydrodissection and post-placement ultrasound. Catheters were secured with adhesive dressings and connected to programmed intermittent bolus pumps delivering ropivacaine.
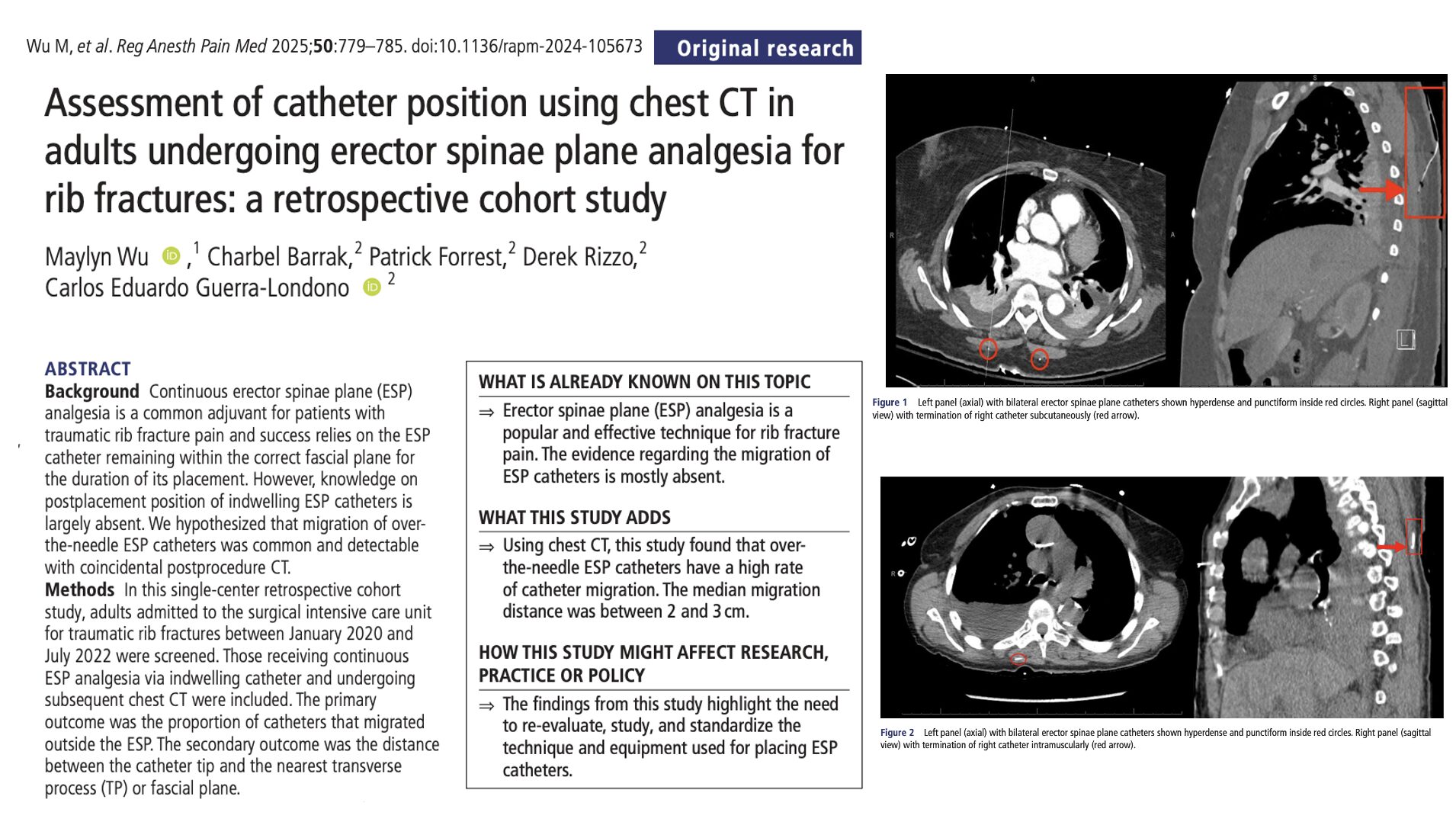
- CT evaluation: Two blinded investigators independently reviewed axial and sagittal CT images to classify catheter position (in-plane, intramuscular, or subcutaneous) and measure the shortest axial and sagittal distances between the catheter tip and the ESP/transverse process. Discrepancies were adjudicated by a third reviewer.
- Primary outcome: Proportion of ESP catheters located outside the intended erector spinae plane.
- Secondary outcomes: Sagittal and axial distance (mm) between catheter tip and ESP; catheter depth changes over time; documented catheter-related issues (migration, dysfunction, insertion-site symptoms); and daily pain scores during the first 5 postoperative days.
Key findings
-
Most ESP catheters were outside the intended fascial plane
Sixteen of the eighteen indwelling ESP catheters were no longer positioned within the erector spinae plane on CT imaging. The majority of catheter tips were located superficial to the plane: fourteen were intramuscular and four were subcutaneous. These findings indicate that catheter migration is extremely common and often substantial despite ultrasound-confirmed placement.
-
Catheter migration averaged 2–3 cm away from the ESP
For misplaced catheters, the median distance between the catheter tip and the ESP was 23.2 mm sagittally and 25.1 mm axially. In several cases, displacement approached 5 cm. These distances suggest that migrated catheters are unlikely to deliver local anesthetic consistently into the ESP, potentially diminishing the benefits of continuous infusion.
-
Clinical assessment did not reliably detect malposition
Despite the high malposition rate on CT, routine bedside assessments rarely document catheter displacement or dysfunction. Only three catheters showed changed skin depth measurements, and only one case of inadequate analgesia was linked to malposition. Skin exams and charted depth, therefore, appear unreliable for detecting ESP catheter migration, which occurs silently and without visible external cues.
-
Catheter-over-needle design and mechanical factors may contribute to migration
The study highlights several likely contributors: the rigidity and limited adjustability of over-the-needle catheter systems, continuous movement of the overlying trapezius and erector spinae muscles, repeated patient repositioning in the ICU, and practices such as vibrating beds for respiratory therapy. These factors may create subtle forces that gradually shift the catheter tip away from its original fascial location.
-
Pain scores improved despite malposition, complicating interpretation
Rest pain scores decreased over the days following placement, reflecting natural recovery, multimodal systemic analgesia, and possibly partial benefit from ESP boluses even when delivered into muscle or subcutaneous layers. This clinical improvement may mask catheter migration, contributing to under-recognition of malpositioned ESP catheters.
Conclusion
In adults receiving continuous ESP analgesia for traumatic rib fractures, most over-the-needle catheters did not remain in the intended fascial plane when assessed on chest CT within 120 hours of placement. Migration typically occurred superficially, averaging 2–3 cm, potentially compromising the efficacy of intermittent bolus delivery. Because catheter malposition was seldom clinically apparent, reliance on skin markings or clinical exam is insufficient in detecting displacement. These findings call for reassessment of ESP catheter techniques, equipment selection, and practice standardization to reduce migration and improve reliability.
Future research
- Prospectively evaluate ESP catheter migration with standardized ultrasound or CT (with or without contrast) at predefined time points.
- Compare CON versus CTN systems, different insertion angles, and varying threading distances beyond the needle tip.
- Link catheter position to objective analgesic outcomes, respiratory metrics, and opioid use.
- Develop and test standardized ESP catheter protocols—potentially including anchoring methods or tunneling—to minimize migration.
- Explore whether alternative techniques (paravertebral, serratus anterior, intercostal catheters) offer more reliable catheter stability in specific trauma populations.
Clinical implications
For clinicians using continuous ESP catheters in rib fracture pathways, this study provides an important reminder that ultrasound-confirmed placement does not guarantee stable positioning over time. Catheter migration appears common, often considerable, and largely undetectable by bedside exam. Persistent or worsening pain, diminished respiratory effort, or unexpected bolus failures should raise immediate suspicion for catheter malposition rather than being attributed solely to injury severity or opioid needs. These findings support careful reassessment of insertion technique, catheter system choice, and securing strategies, as well as maintaining robust multimodal analgesia in case continuous ESP delivery becomes unreliable.
Clinical pearls
- On CT, 16 of 18 ESP catheters were no longer in the fascial plane.
- Median migration distance was about 2–3 cm away from the ESP (up to ~ 5 cm).
- Most tips were intramuscular or subcutaneous, not deep.
- Skin depth and dressing inspection were poor predictors of actual catheter position.
- Pain improved over days, but catheter migration may quietly undermine continuous block efficacy.
Practical tip: If a patient with an ESP catheter for rib fractures shows unexpected pain or poor respiratory effort, assume catheter migration is possible—don’t hesitate to reassess, re-bolus under ultrasound, or replace the catheter.
For more detailed information, refer to the full article in RAPM.
Wu M. et al. Assessment of catheter position using chest CT in adults undergoing erector spinae plane analgesia for rib fractures: a retrospective cohort study. Reg Anesth Pain Med. 2025;50:779-785.
Download the Nerve Blocks App HERE for in-depth insights on the erector spinae plane block. Prefer a physical copy? The bestselling NYSORA Nerve Blocks App is available in book format — an essential resource for mastering nerve blocks! And for a digital learning experience, check out the Nerve Blocks Module on NYSORA360!