Peripheral nerve blocks remain a cornerstone of multimodal analgesia in pediatric surgery, offering targeted, opioid-sparing pain control. Among these, the ultrasound-guided popliteal sciatic nerve block is commonly used for lower-extremity procedures, such as foot and ankle surgery. While long-acting agents like ropivacaine are preferred for their safety and duration, the search for adjuncts that further prolong analgesia without increasing risk continues.
A recent randomized, double-blind, placebo-controlled trial conducted at Poznan University of Medical Sciences and published in Regional Anesthesia and Pain Medicine brings new clarity to this issue. The study evaluated the effectiveness and safety of adding perineural dexamethasone at two different doses to ropivacaine in pediatric sciatic nerve blocks. The findings confirm that dexamethasone significantly extends analgesia duration and reduces opioid use, without influencing systemic metabolic or inflammatory markers.
Study design and methodology
This single-center, triple-arm RCT included 90 children aged 2–5 years undergoing elective foot or ankle surgery under spinal anesthesia. All patients received a standardized ultrasound-guided popliteal sciatic nerve block with 0.2% ropivacaine. Participants were randomized into three groups:
- Control: ropivacaine + saline
- DEX0.05: ropivacaine + 0.05 mg/kg dexamethasone
- DEX0.1: ropivacaine + 0.1 mg/kg dexamethasone
The total volume of injectate was consistent across all groups, and dexamethasone was preservative-free to avoid neurotoxic risk. The primary outcome was time to first rescue opioid analgesia. Secondary outcomes included motor block duration (time to first toe movement), total opioid consumption, pain scores (FLACC), and biochemical markers of surgical stress—neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and blood glucose.
Analgesic duration and motor block
The results demonstrated a clear, dose-dependent effect of dexamethasone on prolonging postoperative analgesia:
- Time to first rescue opioid:
- DEX0.1: 18.4 hours
- DEX0.05: 16.3 hours
- Control: 8.5 hours
- Motor block duration (toe movement):
- DEX0.1: 17.3 hours
- DEX0.05: 15.2 hours
- Control: 7.8 hours
Both dexamethasone groups showed significantly longer sensory and motor block durations compared to the control. The higher 0.1 mg/kg dose extended analgesia by nearly 10 hours compared with saline and by over 2 hours compared with the lower dose.
Opioid consumption and pain control
The use of dexamethasone also translated to a meaningful reduction in opioid requirements:
- Total nalbuphine consumption:
- DEX0.1: 0.53 mg/kg
- DEX0.05: 0.77 mg/kg
- Control: 1.10 mg/kg
Fewer patients in the DEX0.1 group required rescue opioids compared to the control group. Although opioid use in the DEX0.05 group was lower than that of the control, the difference was not statistically significant.
Pain scores, measured using the FLACC scale at multiple postoperative time points, were significantly lower in the dexamethasone groups during the first 20 hours. The greatest differences were observed at 8, 12, and 16 hours post-op, consistent with the extended analgesic duration.
Metabolic and inflammatory markers
One of the most notable aspects of the trial was its evaluation of systemic safety, particularly in very young children. Blood glucose levels, as well as markers of surgical stress (NLR and PLR), remained stable across all groups before surgery and at 24 and 48 hours postoperatively. This suggests that perineural dexamethasone at doses up to 0.1 mg/kg does not increase surgical stress or alter metabolic responses in pediatric patients.
Mechanistic insight
The analgesic-enhancing effects of dexamethasone likely arise from its action on glucocorticoid receptors within peripheral nerves. These actions include modulation of ion channels, suppression of C-fiber activity, and inhibition of local inflammation. By decreasing neuronal excitability, dexamethasone can enhance and prolong the effects of local anesthetics like ropivacaine.
Unlike other adjuvants such as dexmedetomidine, which may cause sedation, bradycardia, or hypotension, dexamethasone is well tolerated and lacks significant hemodynamic side effects—making it particularly suitable for pediatric use when administered perineurally.
Broader context and safety
This trial is among the first to systematically evaluate perineural dexamethasone in children as young as two years old. Previous pediatric data had largely focused on older children or caudal blocks. The current findings are consistent with those from adolescent and adult studies, showing enhanced block duration and reduced pain without systemic complications.
The use of preservative-free dexamethasone, a standardized injection protocol, and ultrasound guidance helped mitigate risks such as nerve injury or systemic toxicity. No adverse events, including nerve deficits or signs of neurotoxicity, were reported in any group.
Implications for clinical practice
This study supports the integration of perineural dexamethasone into pediatric regional anesthesia protocols, particularly in lower limb surgeries with moderate to high expected postoperative pain. The data suggest that 0.1 mg/kg offers the most benefit, with a favorable safety profile even in very young children.
Anesthesiologists aiming to reduce perioperative opioid exposure in children while maximizing the efficacy of peripheral nerve blocks may consider routine use of dexamethasone as an adjuvant in popliteal sciatic blocks—provided the preparation is preservative-free, and the technique is ultrasound-guided.
Conclusion
Perineural dexamethasone at doses of 0.05 mg/kg and 0.1 mg/kg significantly prolongs the duration of ropivacaine sciatic nerve blocks in children undergoing foot or ankle surgery. The higher dose provides superior analgesia and longer motor block without increasing blood glucose levels or systemic inflammatory markers. These findings offer robust support for the use of dexamethasone as an effective and safe adjuvant in pediatric regional anesthesia.
Clinical pearls
- Dexamethasone 0.1 mg/kg nearly doubles analgesia duration vs. ropivacaine alone
- Motor block and pain scores improved in both dexamethasone groups
- No effect on blood glucose, NLR, or PLR—confirming systemic safety
- No nerve injuries or adverse events reported
- Use only preservative-free dexamethasone with an ultrasound-guided technique
For more information, refer to the full article in RAPM.
Reysner M et al. Dexamethasone as a perineural adjuvant to a ropivacaine popliteal sciatic nerve block for pediatric foot surgery: a randomized, double-blind, placebo-controlled trial. Reg Anesth Pain Med. 2025 Dec 5;50(12):970-976.
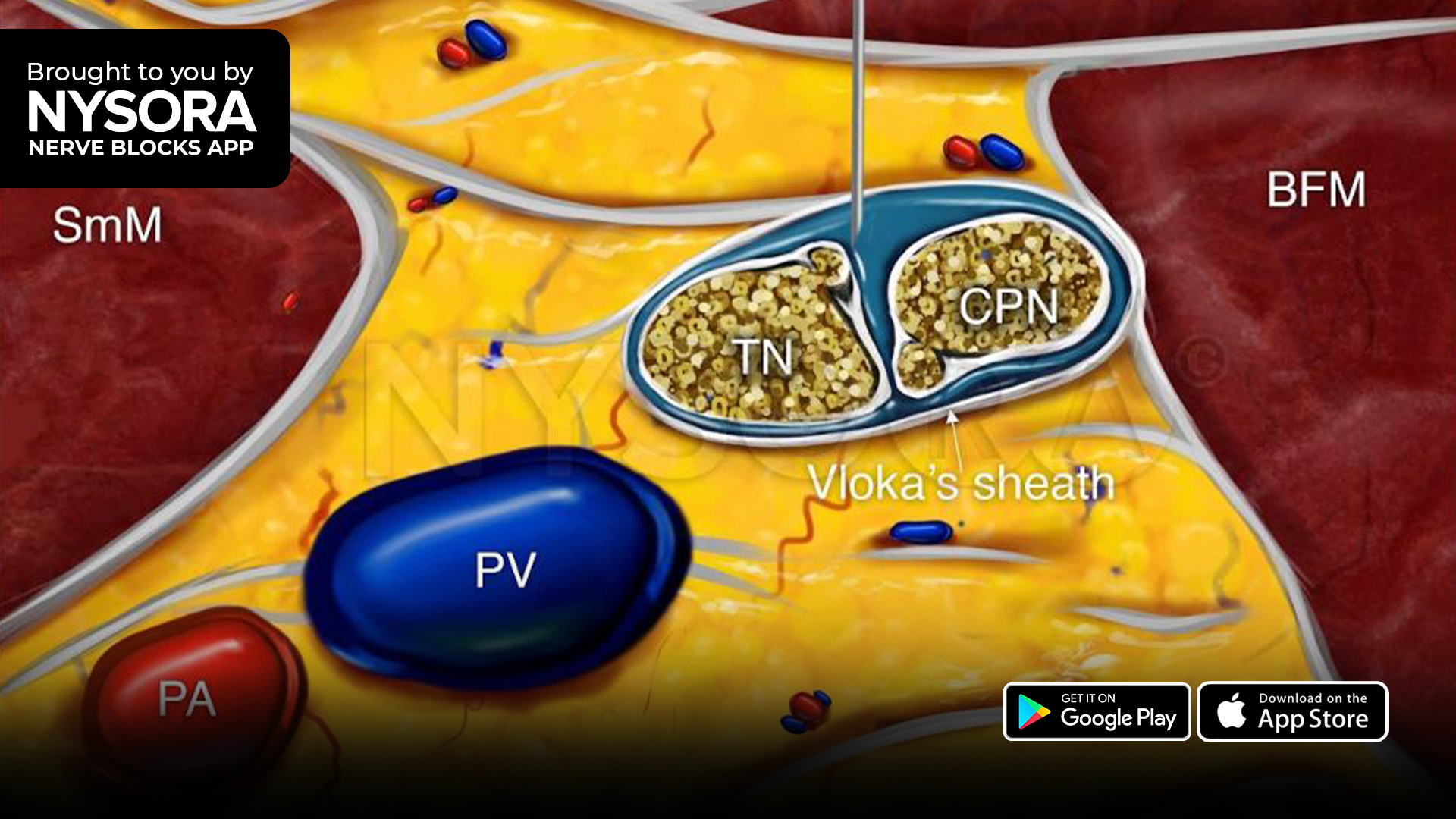
Get full access to the poplitea sciatic nerve block in the NYSORA Nerve Blocks App: technique walkthroughs, PRO tips, animations, and more.