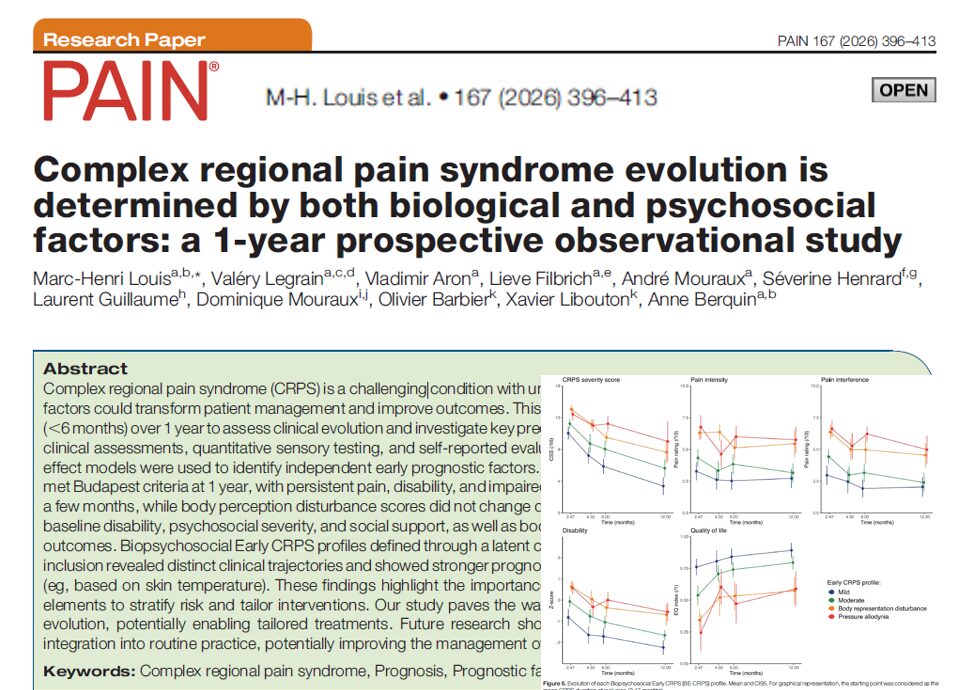
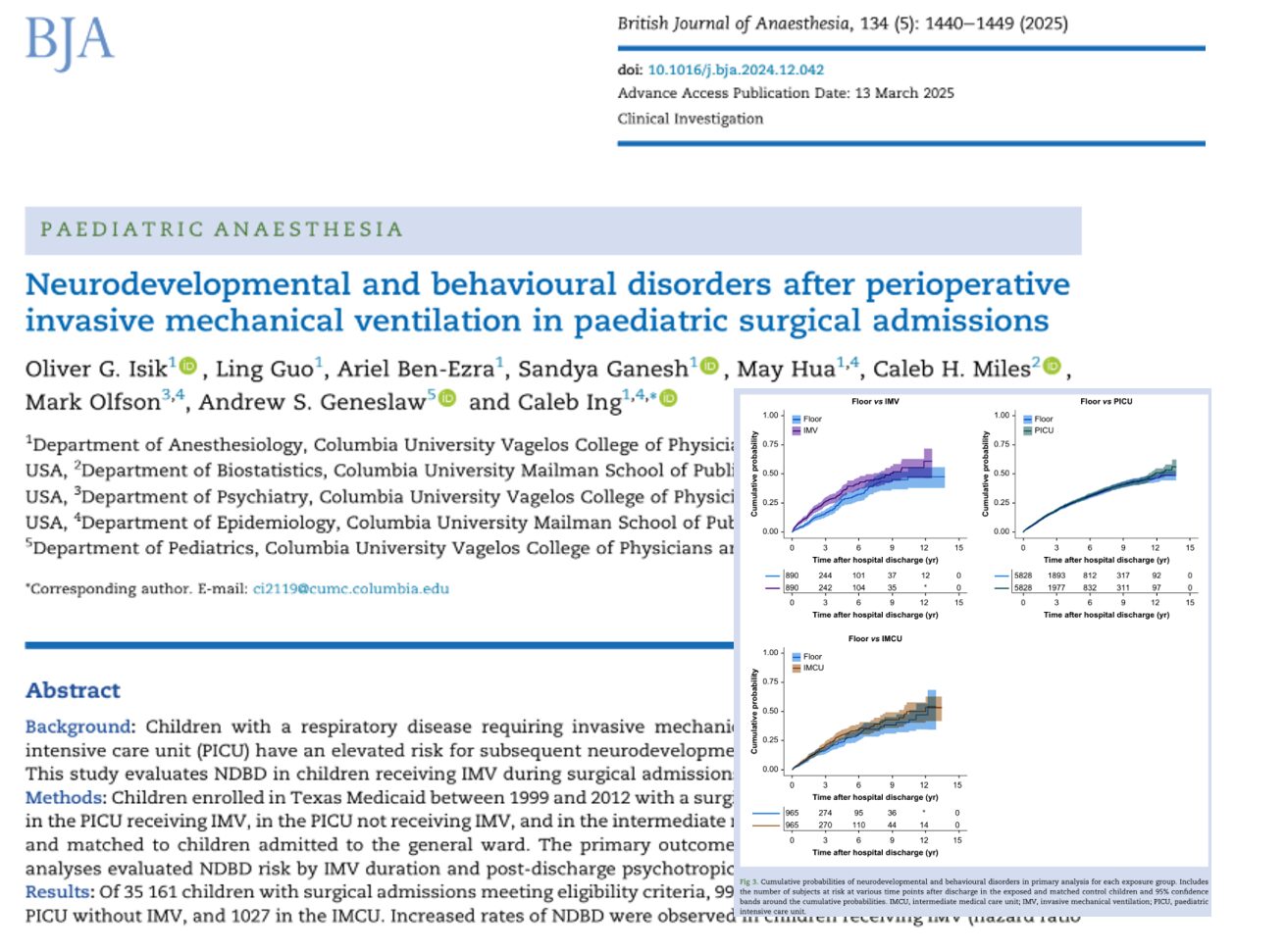
Extracorporeal membrane oxygenation (ECMO) has become a life-saving intervention for patients in severe cardiopulmonary failure. However, its use introduces a complex paradox: while life is sustained through mechanical circulatory support, the very system meant to heal disrupts the body’s natural balance of clotting and bleeding.
Understanding how to manage anticoagulation and hemostasis in ECMO is critical for improving outcomes and reducing complications such as stroke, hemorrhage, and thrombosis.
What is ECMO, and why does it complicate hemostasis?
Extracorporeal membrane oxygenation (ECMO) is a cardiopulmonary bypass technique used in critically ill patients to support heart and/or lung function. The ECMO circuit diverts blood outside the body to oxygenate it and remove carbon dioxide before returning it to the patient.
The hemostatic challenge
ECMO exposes blood to artificial surfaces, high shear stress, and inflammatory stimuli, which:
- Activate the coagulation cascade
- Damage or activate platelets
- Disrupt endothelial homeostasis
- Accelerate fibrinolysis and hemolysis
This results in what’s known as ECMO-associated coagulopathy, a paradoxical state of both increased bleeding and thrombotic risk.
Bleeding and thrombosis: the most frequent complications
According to registry data from the Extracorporeal Life Support Organization (ELSO), up to 50% of ECMO patients experience either bleeding or thrombosis, and many experience both.
Bleeding complications
- Occurs in ~30% of ECMO patients
- More common in venoarterial (V-A) ECMO than in venovenous (V-V)
- Common sites:
- Cannulation sites
- Surgical wounds
- Pulmonary and intracranial hemorrhage
- Cannulation sites
- Risk factors:
- Thrombocytopenia
- Hypofibrinogenemia
- Acidosis
- Surgical cannulation
In one study, early major bleeding in V-A ECMO patients was linked to increased mortality (OR 2.54).
Thrombotic complications
- Occur in 20–60% of ECMO patients, varying by population
- Include:
- Circuit thrombosis
- Pulmonary embolism
- Ischemic stroke
- Cannula-associated DVT (up to 53.5% in V-V ECMO)
- Circuit thrombosis
- Associated with:
- Longer ECMO duration
- COVID-19 infection
- Mechanical circuit damage
- Longer ECMO duration
Despite high incidence, not all thrombotic events increase mortality; pulmonary embolism and ischemic stroke are the exceptions.
The pathophysiology of ECMO-induced coagulopathy
The disruption of hemostasis in ECMO involves several overlapping mechanisms.
-
Contact activation and protein adsorption
Blood contact with artificial surfaces triggers:
- Vroman effect – rapid deposition of proteins like fibrinogen and albumin
- Activation of Factor XII → Thrombin generation
- Formation of a procoagulant surface on the ECMO circuit
-
Platelet dysfunction
- Thrombocytopenia in >90% of V-A ECMO patients
- Loss of platelet activation receptors (e.g., CD62P, CD63, GPIIb/IIIa)
- Formation of platelet-neutrophil aggregates
- Increased clearance of dysfunctional platelets
Platelet loss and dysfunction are critical contributors to bleeding risk, especially during the early days of ECMO support.
-
Acquired von Willebrand syndrome (AVWS)
- Caused by shear-induced cleavage of VWF multimers
- Diagnosed when VWF:RCo/VWF:Ag < 0.7
- Not responsive to desmopressin
- Best treated with VWF concentrates
-
Hyperfibrinolysis
- Elevated tPA, D-dimers, and plasmin activity
- Leads to clot breakdown and impaired hemostasis
-
Hemolysis
- Shear stress causes red cell destruction
- Free hemoglobin activates endothelial cells and coagulation
- Amplifies both thrombosis and fibrinolysis
Current anticoagulation strategies
-
Unfractionated heparin (UFH)
- Most widely used
- Monitored using:
- aPTT
- Anti-Xa levels
- aPTT
- Limitations:
- Variable pharmacokinetics
- Heparin-induced thrombocytopenia (HIT)
- Dependence on antithrombin III
- Variable pharmacokinetics
-
Direct thrombin inhibitors (DTIs)
- Bivalirudin is the most studied
- Advantages:
- Predictable effect
- No antithrombin dependency
- Predictable effect
- Limitations:
- No antidote
- Limited availability
- Higher cost
- No antidote
Meta-analyses show bivalirudin may reduce thrombosis without increasing bleeding, but evidence remains mostly retrospective.
Monitoring coagulation on ECMO
Monitoring strategies remain heterogeneous.
Common modalities
- aPTT (target 60–90 seconds)
- Anti-Xa assay (target 0.3–0.5 U/mL)
- Activated clotting time (ACT) – less specific, more variable
Advanced tools
- Thromboelastography (TEG)
- Rotational thromboelastometry (ROTEM)
- Platelet function tests
These emerging tools help tailor anticoagulation more precisely to each patient’s coagulation status, potentially reducing both bleeding and clotting complications.
Transfusion goals in ECMO patients

Step-by-step: Managing anticoagulation in ECMO
- Initiate UFH upon ECMO cannulation.
- Set aPTT or anti-Xa targets based on patient risk profile.
- Monitor platelets, fibrinogen, and VWF daily.
- Use viscoelastic testing if available to fine-tune therapy.
- Switch to bivalirudin in case of HIT or UFH resistance.
- Adjust targets for patients with high bleeding or thrombotic risk.
- Decannulate cautiously, screening for thrombosis post-ECMO.
Conclusion
Anticoagulation and hemostasis in ECMO patients demand a nuanced, individualized approach. As technologies and understanding evolve, there is promise in reducing ECMO-related complications through more refined monitoring, advanced circuit design, and personalized therapeutic strategies.
The future of ECMO lies not just in supporting life but in mastering the balance between bleeding and clotting that keeps that life viable.
Reference: Saura O et al. Anticoagulation and hemostasis in the extracorporeal membrane oxygenation patient. Curr Opin Anaesthesiol. 2026;39:39-45.
Learn more about anticoagulation guidelines in NYSORA’s Anesthesia Assistant App.