Based on: “Vloka JD, Hadzic A, Mulcare R, Lesser JB, Koorn R, Thys DM. Combined blocks of the sciatic nerve at the popliteal fossa and posterior cutaneous nerve of the thigh for short saphenous vein stripping in outpatients: An alternative to spinal anesthesia. J Clin Anesth 1997;9:618-22”
Abbreviations:
SSVS – short saphenous vein stripping
PNBs – peripheral nerve blocks
Pop NB – popliteal nerve block
PCNTB – posterior cutaneous nerve of the high block
OR – operating room
PACU – post-anesthesia care unit
SSU – short stay unit
|
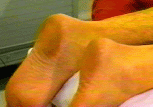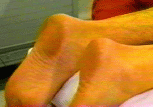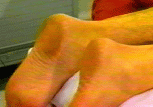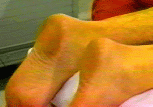
Background 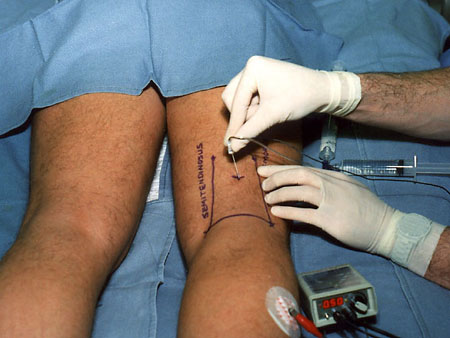 Varicose veins of the lower extremity are a common and progressive disease that, if untreated, often lead to thrombophlebitic, inflammatory, and stasis complications. The most common surgical treatment includes stripping of the long and/or short saphenous veins [1]. In the United States, approximately 46,000 lower extremity varicose vein stripping procedures are performed annually on an outpatient basis [2]*. Since these procedures are associated with relatively minor surgical trauma, the speed of recovery from anesthesia greatly influences the time to discharge from the hospital. Although the operation can be performed under neuraxial or general anesthesia, both of these techniques are associated with some side-effects that may be undesirable in the outpatient setting [3,4,5,6]. Short saphenous vein stripping (SSVS) is performed in the prone position, which entails special considerations particularly with spinal or general anesthesia. While the procedure could also be performed under local anesthesia, this may require significant supplemental intravenous sedation with the patient in the prone position [7,8]. In this study we tested the hypothesis that peripheral nerve blocks will provide adequate anesthesia, faster recovery and better postoperative pain control than spinal anesthesia for outpatients undergoing SSVS. Methods After obtaining institutional review board approval and patients’ informed consent, patients of ASA physical status I-II undergoing SSVS were randomized to receive peripheral nerve blocks or spinal anesthesia. The randomization was performed by the method of sealed envelopes immediately prior to surgery. All patients received midazolam 1-2 mg, and some received fentanyl 0-100 mcg intravenously just prior to performance of the blocks. Additional doses of midazolam and fentanyl were administered during the operation as necessary to allay anxiety or discomfort unrelated to the surgical instrumentation (positioning, discomfort during blood pressure checking, etc.). In case of specific complaints related to the surgical procedure (i.e., pain on instrumentation), infiltration with local anesthetic was used instead of intravenous supplementation.  Figure 1. Popliteal nerve block: Posterior approach. The needle attached to a nerve stimulator is inserted between the tendons of the semitendinosus and biceps femoris muscles. Patients assigned to the peripheral nerve blocks group received a popliteal nerve block (Pop NB) and posterior cutaneous nerve of the thigh block (PCNTB) (Figure 1). Pop NB was performed with patients in the prone position [8] using a 22G gauge 50 mm, Teflon coated needle (Stimuplex®, B. Braun Medical Inc, Bethlehem, PA). A peripheral nerve stimulator stimulator (DualStim®; DX, Professional Instruments, Houston, TX), with the negative electrode connected to the needle and the reference electrode connected to the lateral calf, was used to localize the popliteal nerve. After an appropriate response to nerve stimulation was obtained (either plantar or dorsal flexion of the foot was sought at a stimulating current of 0.5 mA or less), 30 mL of 3% alkalinized chloroprocaine were injected. The solution of chloroprocaine was alkalinized by adding 1 mEq of sodium bicarbonate to 30 mL commercially prepared 3% chloroprocaine solution (Abbott Labs, Chicago, IL). PCNTB was performed also in the prone position by subcutaneous infiltration with 10 mL of 1% lidocaine along a line 5 cm proximal and parallel to the popliteal crease.
Spinal anesthesia was induced using the midline approach with the patient in the sitting position and aiming for the L3-4 interspace. Following infiltration of the skin with 3-5 mL of 1% lidocaine, a 24-gauge, 90 mm spinal needle (Pencil point, Gertie MarxTM Spinal Needle, IMD, Inc. Park City, UT) was inserted through a 20 gauge introducer in the sitting position. After aspirating approximately 1 mL of cerebrospinal fluid, 65 mg of 5% hyperbaric lidocaine through a 24G Sprotte needle were injected. Seven minutes later, after allowing for the block to “set-in,” the patients were positioned in the prone position. All patients received intravenous midazolam 1-2 mg and fentanyl 50 mcg, in incremental doses of 0.5 mg and 25 mcg respectively, prior to the introduction of the anesthetic. The need for additional sedation or analgesia was adjusted for patient comfort as necessary. All anaesthetics were administered in the operating room (OR), and all procedures were performed by the same surgeon, using identical surgical techniques. With the patient in the prone position, a 1-2 cm incision was made over the sapheno-popliteal vein junction. The short (lesser) saphenous vein was identified beneath the popliteal fascia and its branches ligated. The vein was then ligated at the sapheno-popliteal junction, and an internal stripper was passed and retrieved through an incision in the mid-distal leg. After ligation of the distal lesser saphenous vein, its proximal portion was stripped and the wound was closed. Following the operation, the patients were transferred to the post-anesthesia care unit (PACU; Phase 1 recovery) where, according to the institutional protocol, they were assessed immediately upon admission and every 15 minutes thereafter. This protocol also mandated a stay of 60 minutes in the PACU following the operation. Recovery was assessed using a post-anesthetic recovery score (Aldrete score) [9] and clinical criteria (stable vital signs, ability to sit up without dizziness and/or nausea, alert and oriented, tolerable or no pain). Once the patients achieved a post-anesthetic discharge score of 9 or more, they were transferred to the short stay unit (SSU; Phase 2 recovery). Discharge readiness (home readiness) from the SSU was assessed every 30 minutes. Patients were discharged home after having met the following criteria: stable vital signs for at least 1 hour, ability to ambulate and dress with minimal assistance, ability to tolerate liquids orally with minimal nausea and vomiting, and ability to void. Morphine sulfate IV (1 to 2 mg every 5 to 10 minutes) was used for treatment of pain in the PACU, while acetaminophen 300 mg with codeine 30 mg orally was administered in the SSU if the patients complained of pain. Upon discharge from the hospital, the patients were instructed to take one tablet of acetaminophen 300 mg with codeine 30 mg every 3-4 hours as needed for pain. Decisions regarding transfer from the PACU to SSU, discharge home, and the need for pain treatment were made by anesthesiologists and nurses blinded to the study goals. Data collected included: patient demographics, postoperative complications, degree of postoperative discomfort, incidence of pain requiring therapy, requirement for analgesics in the hospital and at home during the first postoperative 24 hours. Study patients were called 24 hours after surgery by a physician blinded to the type of anesthesia that was administered. Their responses to questions hours, were also collected. Patients were called at their homes 24 hours after discharge from the hospital. Responses to questions regarding their experiences during block placement, surgery, recovery, postoperative pain and development of any complication were recorded by a physician blinded to the type of anesthesia. Specific questions were also asked to elicit any symptoms of backache, headache, leg paresthesias and the amount of analgesics taken at home during the first 24 postoperative hours. Patient satisfaction with the anesthetic technique was evaluated during the same telephone interview. by asking the patients whether they would choose the same type of anesthesia for another operation or recommend it to their friends. Time intervals were defined as follows:
Statistical analysis was performed using SPSS (statistical software package for windows, V. 5.02, 1993, Chicago, IL) and included Student’s t and chi-square tests. P values less than 0.05 were considered significant. Results Twenty-eight patients, mean age 53 +/- 3.2 years, were enrolled in the study. Fourteen patients received PNBs, and 14 patients received spinal anesthesia. None of the patients were lost for follow-up. There were no significant differences in the demographic characteristics of the groups (Table 1). There were no failures to achieve the blocks. There were no significant differences in the amount of intravenous sedatives administered intraoperatively between the two groups. Patients in the PNBs group received more fentanyl than patients who were assigned to spinal anesthesia (44.6 +/- 34.2 mcg vs. 15.4 +/- 30.2 mcg respectively, p= 0.02). The use of PNBs was associated with a faster recovery and earlier discharge home (Table 2). Nine patients (64%) who received spinal anesthesia had postoperative pain requiring treatment in the hospital compared to only three patients (21%) who received PNBs (p=0.02). There was no statistically significant difference in the need for pain treatment at home between the two groups (8 or 57.1% in the spinal anesthesia group vs. 5 or 36.0% in the PNBs group).
Table 2:Time requirements of patients undergoing SSVS under PNBs or spinal anesthesia.
The only complications noted during the follow-up phone interview were transient backache (two patients in spinal anesthesia group, one in the PNBs group) and mild headache (one patient in each group). None of the patients complained of residual paresthesias or weakness in the lower extremities. All patients expressed good overall satisfaction with both anaesthetic techniques. Discussion In our series, the PNBs using short acting local anesthetics provided excellent anesthesia and postoperative analgesia in outpatients undergoing SSVS, and resulted in more favorable recovery profile compared to that of spinal anesthesia with lidocaine. 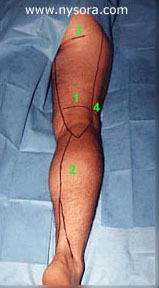
Figure 2: Sensory innervation of the leg. Legend: 1.Posterior Cutaneous nerve of the thigh; The onset of PNB and PCNTB was fast, allowing the surgery to start immediately after the skin preparation was completed (anesthesia onset time 10.4 +/- 5.8 minutes). Using 3% chloroprocaine for Pop NB resulted in fast onset of anesthesia with short lasting motor block, which rapidly regressed by the end of surgery. The residual sensory block in the distribution of the popliteal nerve and posterior cutaneous nerve of the thigh provided excellent postoperative analgesia in the majority of patients. This frequently resulted in no need for analgesics in the PACU and SSU (79% of patients), while allowing early ambulation. This is in contrast to spinal anesthesia with 5% lidocaine that invariably resulted in more prolonged and pronounced motor and proprioception block (longer stay in PACU), and shorter lasting postoperative analgesia (64% of patients required pain treatment prior to discharge home). The proximal incision for SSVS takes place above the popliteal fossa crease in the area innervated by the posterior cutaneous nerve of the thigh. Because this cutaneous branch leaves the sciatic trunk high in the thigh, the popliteal nerve block does not result in anesthesia of the skin covering the popliteal fossa. Since anesthesia of the thigh is not needed for SSVS, we blocked the popliteal fossa (Figure 2), instead of at the gluteal crease. This modification of the previous description of the PCNTB [10] reliably blocks distal branches of the nerve, and avoids unnecessary anesthesia of the posterior thigh. Another advantage of using PNBs for this operation is that the anesthesia is induced in the prone position, which is the same position in which the surgery is performed. This allows for an expeditious start of surgery, and avoids the hazards of spinal anesthesia with patients in the prone position. In summary, the combination of PNB and PCNTB using short acting local anesthetics provides excellent anesthesia for SSVS in outpatients. It results in fast onset of anesthesia, predictable recovery, and better postoperative analgesia than spinal anesthesia with 5% hyperbaric lidocaine. This technique has low potential for complications and unplanned hospital admissions, and avoids the potential hazards of REFERENCES: 1. Keith LM, Smead WL. Saphenous vein stripping and its complications. Surgical Clinics of North America 1983; 2. Graves EJ. Detailed diagnoses and procedures, National Hospital Discharge Survey, 1992. National Center for Health 3. Fancourt-Smith PF, Hornstein J, Jenkins LC. Hospital admissions from the surgical Day Care Centreer of Vancouver General Hospital 1977-1987. Can J Anaesth 1990; 37: 699-704. 4. Gold BS, Kitz DS, Lecky JH, Neuhans JM. Unanticipated admission to the hospital following ambulatory surgery. JAMA 1989; 262: 3008-10. 5. Osborne GA, Rudkin GE. Outcome after day-care surgery in a major teaching hospital. Anaesth Intensive Care 1993; 21: 822-7. 6. Watcha MF, White PF. Postoperative nausea and vomiting. its etiology, treatment, and prevention. Anesthesiology 1992; 77: 162-84. 7. Georgiev M, Ricci S, Carbone D, Antignani P, Moliterno C. Stab avulsion of the shot saphenous vein. Technique and Duplex examination. J Dermatol Surg Oncol 1993; 19: 456-464. 8. Ricci S, Georgiev M. Office varicose vein surgery under local anaesthesia. J Dermatol Surg Oncol 1992; 18: 55-58. 9. Brown DL. Popliteal Block. In: Brown DL, eds. Atlas of Regional Anaesthesia. Philadelphia: W.B. Saunders Company, 1992; 109-113. 10. Brown TCK, Dickens DRV. A new approach to lateral cutaneous nerve of thigh block. Anaesth Intens Care 1986; 14: 126-127. |
|||||||||||||||||||||||||||||||||||||||||||||||||||