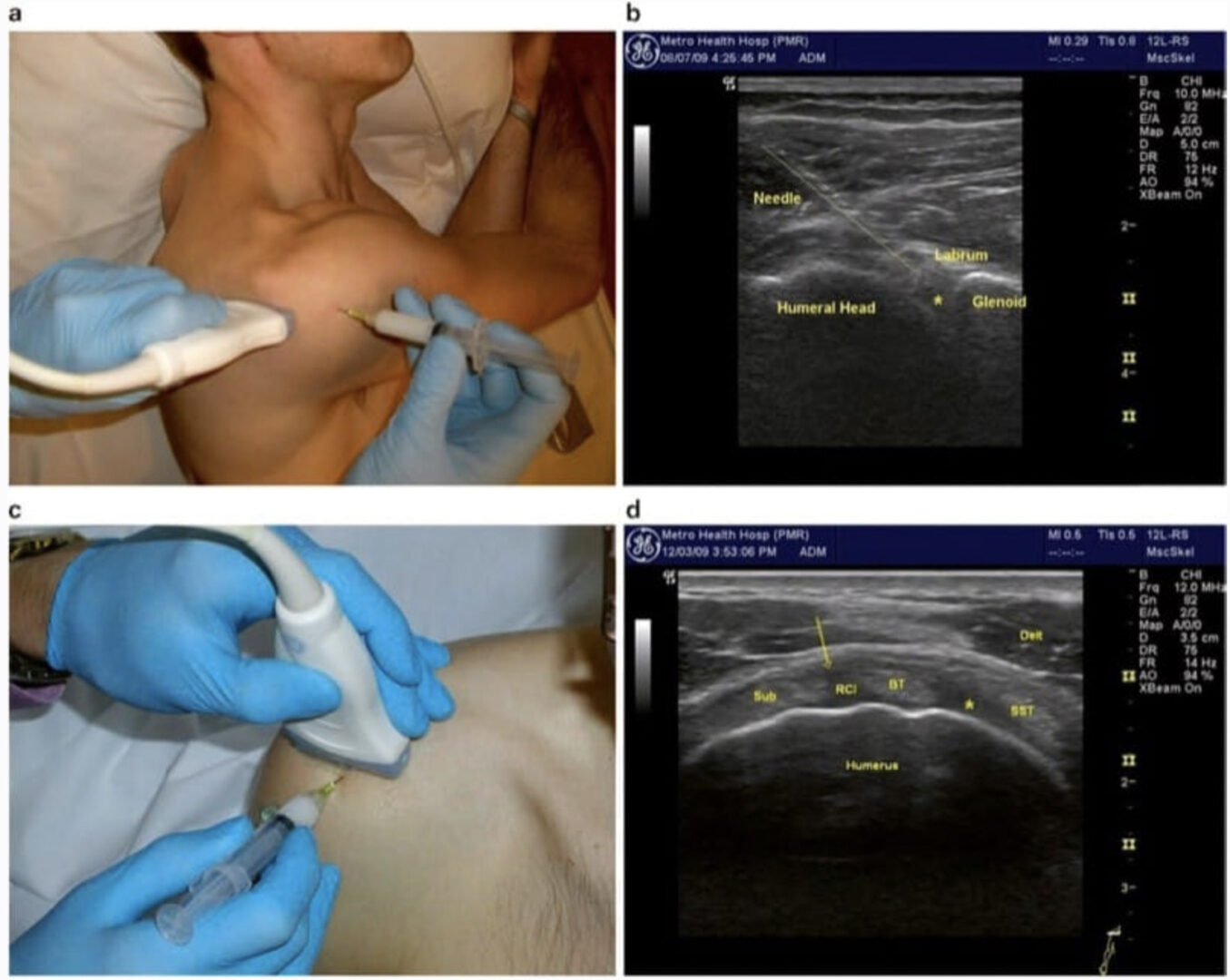
Patients with pain, numbness, and weakness in the upper extremity are frequently referred to pain specialists. Carpal tunnel syndrome (CTS) combined with shoulder impingement can easily mimic cervical radiculopathy and disk herniation. Chronic pain at the thenar eminence following carpal tunnel surgery may stem from occult trigger thumb or carpometacarpal (CMC) joint arthritis. Median neuropathy at the wrist combined with impingement of the flexor pollicis longus (FPL) tendon on a fixation plate screw following fracture of the radius can mimic the pain, burning, and weakness of complex regional pain syndrome (CRPS). These and other conditions of the hand, wrist, and elbow can be effectively diagnosed and treated with diagnostic ultrasonography and ultrasound-guided injections. A few general principles apply with regard to ultrasound-guided injections in the hand, wrist, and elbow. The structures are small and superficial, so a small high-frequency transducer (>12 MHz) is best because of its maneuverability and high resolution. Adequate gel is necessary to maintain good skin contact while scanning over bony structures. The tip of a curved hemostat or other small instrument or the examiner’s little finger can be used to help determine which specific structures are tender, such as the CMC joint of the thumb or the adjacent scaphoid-trapezium-trapezoid (STT) joint. A model of the hand, wrist, and elbow placed next to the patient and ultrasound machine can be useful for teaching purposes and visualization of complex anatomy, such as the bony contours of the carpal bones.
1. ANATOMY ULTRASOUND-GUIDED CARPAL TUNNEL INJECTIONS
The carpal tunnel contains the median nerve and nine tendons, including the flexor digitorum superficialis (FDS), profundus (FDP), and pollicis longus (FPL) (Fig. 1). The tendons are retained by the flexor retinaculum, which extends from the tubercle of the trapezium and scaphoid to the hook of the hamate and pisiform. The FDS and FDP tendons are surrounded by a common synovial sheath, while the FPL has a separate sheath. The location of the median nerve is just beneath the flexor retinaculum, medial to the flexor carpi radialis (FCR), superficial to the FPL, and lateral to the FDS; however, it may be located up to a centimeter or more medially; thus even the best performed blind carpal tunnel injections may injure the nerve. The normal median nerve moves in response to finger movements, which can be seen with dynamic ultrasound imaging.
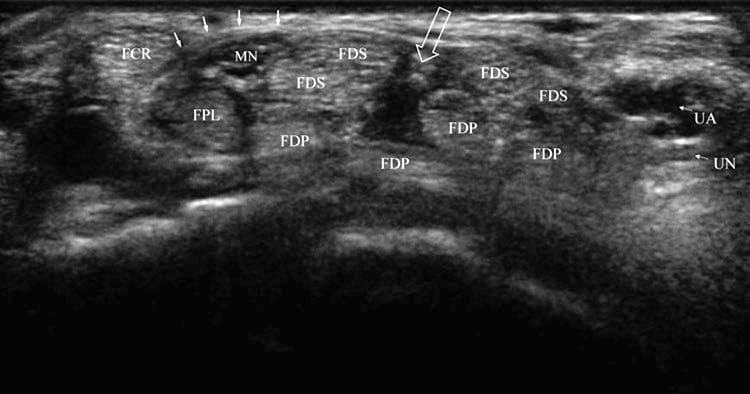
Fig. 1 Normal carpal tunnel. Short-axis view at the distal wrist crease and opening of the carpal tunnel showing typical anatomy in an unaffected individual. The FCR is separated from the median nerve (MN) and FPL by the transverse retinaculum (solid arrows). A cleft or opening (open arrow) is seen between FDS tendons halfway between the median and ulnar nerve (UN) and artery (UA).
CTS is the most common peripheral nerve entrapment syndrome. The symptoms include numbness in the hand at night, pain, weakness, and a feeling that the hand is swollen. Sensation is decreased in the volar aspect of the thumb, index, middle, and radial half of the ring finger. The gold standard for diagnosis remains nerve conduction studies and electromyography, but ultrasound criteria for CTS have been developed and include median nerve cross-sectional area (CSA) at the distal wrist crease >15 mm2, median nerve CSA ratio between distal wrist crease and 12 cm proximally >1.5 (we use >2.0 for greater specificity), and bowing of the flexor retinaculum.
2. LITERATURE REVIEW ON ULTRASOUND-GUIDED CARPAL TUNNEL INJECTIONS
Grassi et al. described a short-axis technique for carpal tunnel injection in a case of CTS caused by rheumatoid synovitis in which the needle was directed into the interval between the median nerve and the FCR tendon. In our experience, this interval is too narrow to allow easy access to the carpal tunnel in most people but is an option when the median nerve is located more medially (Fig. 2).
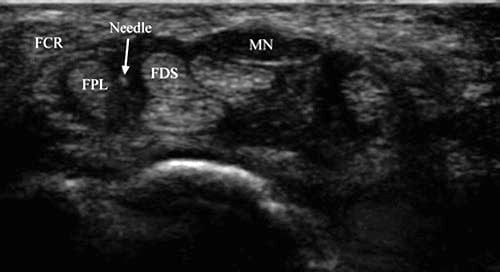
Fig. 2 Carpal tunnel syndrome (short-axis injection) – medially displaced median nerve. The median nerve (MN) is medially displaced, and an opening is present between FPL and FDS tendons allowing passage of the needle (arrow).
Smith et al. described a long-axis ultrasound-guided carpal tunnel injection technique which is performed at the level of the pisiform. The needle is inserted just superficial and lateral to the ulnar nerve and artery and directed toward the median nerve at a shallow angle. Hydrodissection is used to peel the median nerve away from any adhesions. Smith et al. performed over 50 injections using this technique with no complications. The long-axis technique ensures that the needle tip and shaft are seen at all times. We have found this technique to be especially useful in cases of failed carpal tunnel surgery, when injecting directly into the transverse carpal ligament or into the middle of the carpal tunnel where the nerve and tendons are closely packed.
At this time, there are no outcome studies comparing ultrasound-guided vs. blind carpal tunnel injections. A recent review of blind carpal tunnel corticosteroid injections found that 75% of patients treated with carpal tunnel release surgery had excellent outcomes, while 8% got worse. With injections, 70% of patients had excellent short-term outcomes, but 50% relapsed at 1 year.
Armstrong et al. discovered improvement of nerve function, specifically return of absent median sensory nerve action potentials 2 weeks following blind carpal tunnel corticosteroid injections, findings which are of potential significance to all pain specialists, particularly those treating the spine.
The advantage of a short-axis technique is that it deploys the thinnest possible needle at the shortest distance. When performed correctly, it is nearly painless; however, if the needle is jabbed into a tendon, the patient will experience pain. We have used the following technique in over 1800 ultrasound-guided carpal tunnel injections with only one complication (infection in a patient with a previous history of infection).
3. ULTRASOUND-GUIDED TECHNIQUE FOR CARPAL TUNNEL INJECTIONS
The patient is seated across from the pain interventionist with the wrist and hand in supination resting on a pillow. The patient is seated next to the ultrasound machine so that the interventionist does not have to turn his head or significantly alter his gaze, factors which could affect the accuracy of needle placement.
The fingers are flexed, and the hand is relaxed to maximize space between tendons, and then a short-axis view at the distal wrist crease is obtained. An opening between the flexor tendons, usually a vertical or slightly diagonal cleft located halfway between the median and ulnar nerves and most often between the middle and ring finger FDS tendons, is identified (Figs. 1 and 3a–c). When performing an ultrasound-guided injection in either the short or long axis, it is important to remember that the site of needle insertion always lies outside of view of the ultrasound screen. Thus, it is necessary to briefly scan over the intended needle insertion site to make sure any sensitive structures, such as the median or ulnar nerve or artery, are not in the way. The median nerve can be differentiated from the tendons on the basis of their anisotropy or change in appearance from light to dark as the transducer is tipped back and forth in the sagittal plane. It should also be noted that the median nerve can sublux medially or laterally depending on transducer orientation and position.
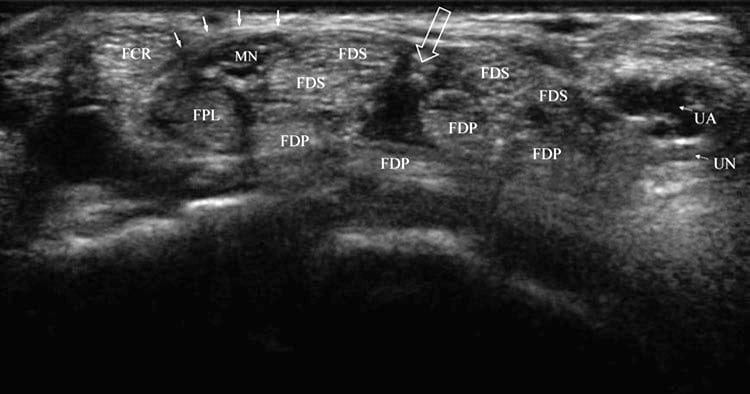
Fig. 1 Normal carpal tunnel. Short-axis view at the distal wrist crease and opening of the carpal tunnel showing typical anatomy in an unaffected individual. The FCR is separated from the median nerve (MN) and FPL by the transverse retinaculum (solid arrows). A cleft or opening (open arrow) is seen between FDS tendons halfway between the median and ulnar nerve (UN) and artery (UA).
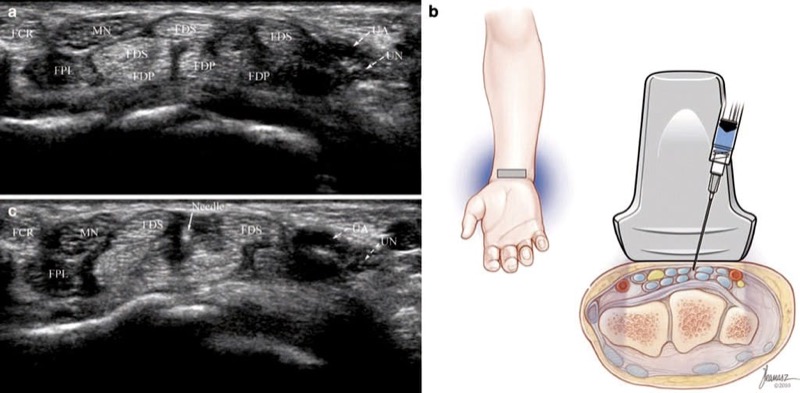
Fig. 3 Carpal tunnel syndrome (short-axis injection). (a) Note enlargement of the median nerve (MN) and the opening between FDS tendons. (b) Illustration showing needle and transducer position prior to injection. Medical illustrations by Joseph Kanasz, BFA. (c) Ultrasound image obtained during injection shows needle tip (arrow) surrounded by anechoic injectate.
After the target is centered on the ultrasound screen, the distance between needle insertion site and the target is calculated using the ultrasound machine caliper tool or estimated on the basis of the scale on the screen. We typically insert a 30-gauge, 25 mm needle in the short axis and slightly obliquely pass through the cleft with minimal to no contact with tendons. We hold the syringe lightly in the hand in order to sense the needle slipping between tendons instead of jabbing into them. When the tip of the needle is within the superficial row of tendons, approximately 1.5 ml of 20–40 mg triamcinolone acetonide and normal saline are injected (Fig. 3b, c). If the medication is not well mixed or the needle jabs into a tendon, clogging may occur and require insertion of another possibly larger gauge needle.
After the needle is withdrawn, the patient is asked to fully extend the fingers, thus drawing medication into the carpal tunnel. Combined with use of a wrist splint at night and avoidance of exacerbating activities, the injection can provide complete relief of symptoms in mild to moderate cases of CTS for up to 6 months or longer in our experience.
4. ANATOMY ULTRASOUND-GUIDED TRIGGER FINGER INJECTIONS
Triggering occurs at the first annular (A1) pulley, where there is an increase in friction or a mismatch in size between the flexor tendons and pulley. The A1 pulley consists of annular bands of connective tissue located at and proximal to the metacarpophalangeal (MCP) joint and contiguous with the tendon sheath. The mean length of the A1 pulley is 12 mm for the adult index, middle, and ring fingers and 10 mm for the little finger. Ultrasound imaging findings of trigger finger include swelling of the tendons, hypoechoic thickening of the A1 pulley, hypervascularization, synovial sheath effusion, and dynamic changes in the shape of the sheath during flexion and extension.
On axial ultrasound views, the A1 pulley is hypoechoic and shaped like an inverted parabola overlying the FDS and FDP tendons and volar plate. In thumbs, the A1 pulley has a more circular shape because of only one tendon present, the FPL.
Trigger finger is a common hand problem, with a lifetime prevalence of 2.6% in the general population and 10% among those with diabetes. Symptoms may range from a vague sense of tightness in the fingers or pain in the palm of the hand to overt triggering and locking. Tenderness is almost always present at the A1 pulley and in mild cases may be the only clue as to the presence of the disorder. Trigger finger can be graded according to the Quinnell scale as follows: 0, normal movement; 1, uneven movement; 2, actively correctable locking; 3, passively correctible locking; and 4, fixed deformity of the digit.
5. LITERATURE REVIEW ON ULTRASOUND-GUIDED TRIGGER FINGER INJECTIONS
Godey et al. published a long-axis technique and demonstrated deposition of steroid below and above the pulley in a single patient. Bodor and Flossman described a short-axis technique in their prospective study of 50 of 52 consecutive trigger fingers, noting complete resolution of symptoms in 94% of fingers at 6 months, 90% at 1 year, 65% at 18 months, and 71% at 3 years. The results were statistically significant and compared favorably to the 56% success rates reported at 1 year for blind injections.
6. ULTRASOUND-GUIDED TECHNIQUE FOR TRIGGER FINGER INJECTIONS
Using the short-axis technique, the target for injection is a triangle under the A1 pulley whose borders consist of the FDS and FDP tendons and volar plate, the distal metacarpal bone, and the pulley (Fig. 4). The flexor tendons are identified in an axial view at the level of the proximal phalanx. At this location, the underlying surface of the bone appears concave. As the transducer is passed more proximally, the concave surface of the proximal phalanx gives way to the convex surface of the metacarpal bone as the MCP joint is crossed.
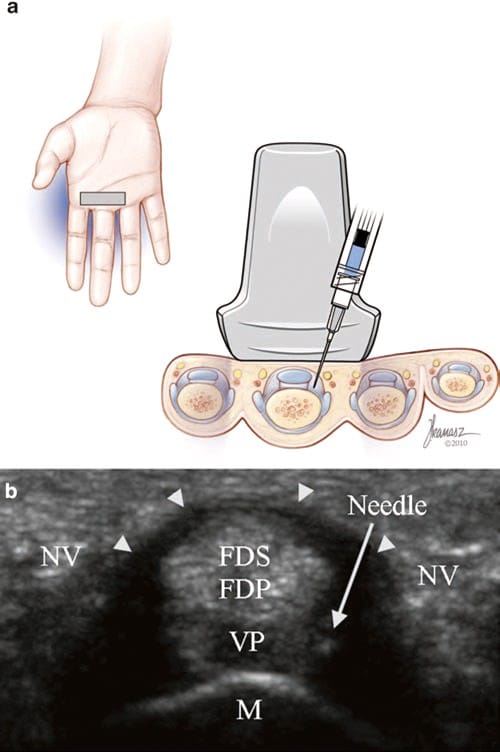
Fig. 4 Trigger finger (short-axis injection). (a) Illustration and medical illustrations by Joseph Kanasz, BFA (b) short-axis view of the A1 pulley (arrowheads) with the tip of the needle inside the target tri- angle, consisting of the A1 pulley (arrowheads), FDS and FDP tendons, volar plate (VP), and distal metacarpal bone (M). The neurovascular bundles (NV) lie on both sides of the pulley.
At this level, the A1 pulley and target triangle are identified and centered on the screen or slightly to the left of center for someone injecting with the right hand. It does not matter whether the triangle on the radial or ulnar side of the tendons is selected. This as well as other short-axis injections requiring such a high degree of accuracy can be facilitated by placing a mark on the side of the transducer indicating its exact center.
We use a distal-to-proximal approach and plan a trajectory to the hypotenuse of the triangle using an approximately 70° angle to horizontal in the axial plane and a 45° angle in the sagittal plane. As soon as the 30-gauge needle punctures the skin, we inject with 0.25 ml of 4% lidocaine for immediate anesthesia and then carefully advance the needle into the target triangle using real-time ultrasound guidance.
When the tip of the needle is inside the triangle, the syringes are switched and approximately 0.5–1.0 ml of 10–15 mg triamcinolone acetonide and lidocaine 2–4% are injected, making sure to visualize flow under the A1 pulley. If flow occurs outside the pulley or there is no flow, the needle is adjusted until flow is obtained. Sometimes initially high resistance to outflow is noted followed by a steep drop in resistance accompanied by visual distention of the pulley. The pulley can be tough to penetrate and the needle may clog, requiring insertion of another possibly larger gauge needle. Afterward, the patient is encouraged to resume usual activities.
7. ANATOMY ULTRASOUND-GUIDED WRIST INJECTIONS
The wrist consists of the distal radius and ulna; the proximal carpal row, including the scaphoid, lunate, triquetrum, and pisiform; the distal carpal row, including the trapezium, trapezoid, capitate, and hamate; and the bases of the metacarpal bones. The wrist joints are grouped as follows: distal radioulnar, radiocarpal, midcarpal, and carpometacarpal. The distal radioulnar joint allows the radius to pivot around the ulna during pronation and supination. The biconcave radiocarpal joint permits both wrist flexion and extension and radial and ulnar deviation. The proximal carpal row serves as a rigid intercalated segment within the wrist kinetic chain and forms a semirigid ring with the distal carpal row. The distal carpal row serves as a solid base of support for the metacarpal bones, and a complex array of ligaments, description of which is beyond the scope of this chapter, connect and stabilize the carpal bones.
The wrist is vulnerable to both acute and chronic injury, including dorsal and volar dislocations, chronic instabilities, rheumatoid and inflammatory arthritides, and osteoarthritis. Osteoarthritis can be classified as primary or secondary. The most common site of primary osteoarthritis in the hand and wrist involves the CMC joint of the thumb. Secondary osteoarthritis typically occurs after fractures or following disruption of the two most important wrist ligaments, the scapholunate and lunotriquetral. Approximately 95% of cases of secondary arthritis involve the scaphoid bone.
8. LITERATURE REVIEW ON ULTRASOUND-GUIDED WRIST INJECTIONS
Koski et al. performed US-guided wrist injections in 50 patients with active rheumatoid arthritis (RA). In the first group, patients were injected with triamcinolone hexacetonide 20 mg entirely into the radiocarpal joint, while in the second group, half the dose was provided to the radiocarpal joint and half to the midcarpal joint. At 3 months, visual analog scores (VAS) improved in both groups, with 19 of 25 wrists in the first group being clinically assessed as better or normal and 22 of 25 in the second group.
Boesen et al. injected the radiocarpal joint of each of 17 RA patients with 1 ml methylprednisolone 40 mg, 0.15 ml gadolinium, and 0.5 ml lidocaine 0.5% with the goal of assessing distribution of contrast among the four wrist compartments. A short-axis approach was used with the transducer sagittally oriented between the distal radius and lunate. A value of 1 was assigned for complete spread within one compartment, 0.5 for partial spread, and 0 for no spread. The mean distribution score was 2.4, with greater distribution noted in patients with higher MRI synovitis scores and distribution in all four compartments noted in only two patients.
In their retrospective study of US-guided contrast injections for MR arthrography, Lohman et al. noted that 101 of 108 (93.5%) injections were intra-articular. Their injection technique involved placing the wrist in slight volar flexion and palpating for Lister’s tubercle. Ultrasound scanning in the short axis was used to identify and mark the space between the third and fourth tendon compartments at the radiocarpal joint; the transducer was rotated 90° and the needle inserted in the long axis.
Umphrey et al. performed US-guided short-axis injections of the trapeziometacarpal (TMC) or thumb CMC joint in cadavers. Fluoroscopic images confirmed intra-articular contrast in 16 of 17 (94%) joints following a single attempt. Mandl et al. reported similar success rates (91%) with blind injections, using ultrasound for confirmation.
In a recent study of 18 patients, Salini et al. provided a single ultrasound-guided injection of sodium hyaluronate 1% to the CMC joint of the thumb, noting at 1 month follow- up a reduction of pain from 1.8 to 0.5 at rest and 8 to 4 with activities, with the elimination of NSAID use in 9 patients and reduction of NSAID use (2.5–1 tablet per week) in 7 patients.
In a well-controlled nonultrasound-guided study of 56 patients with thumb CMC joint arthritis, Fuchs et al. compared one triamcinolone acetonide (TA) 10 mg injection to three 1 ml injections of sodium hyaluronate (SH) 1% given 1 week apart. The VAS score went from 61 to 20 to 48 in the TA group and from 64 to 30 to 28 in the SH group 3 weeks following the last injection and at 26 weeks final follow-up.
9. ULTRASOUND-GUIDED TECHNIQUE FOR WRIST INJECTIONS
A precise sonographic examination is advised before planning any injections. Thus, for example, if treating pain at the radial aspect of the wrist, the radial-scaphoid joint is visualized and centered on the screen and careful palpation performed over the joint to confirm that it is the pain generator. To facilitate precise sonopalpation, we recommend use of a small probe or the tip of one’s little finger. If a specific joint is the pain generator, we expect it to be tender relative to adjacent structures. We find this technique to be especially useful in identifying pain arising from small and difficult-to-access structures such as the pisotriquetral (PT) and STT joints.
Two techniques of wrist injections will be described, the first using a long-axis approach and the second using a short-axis approach.
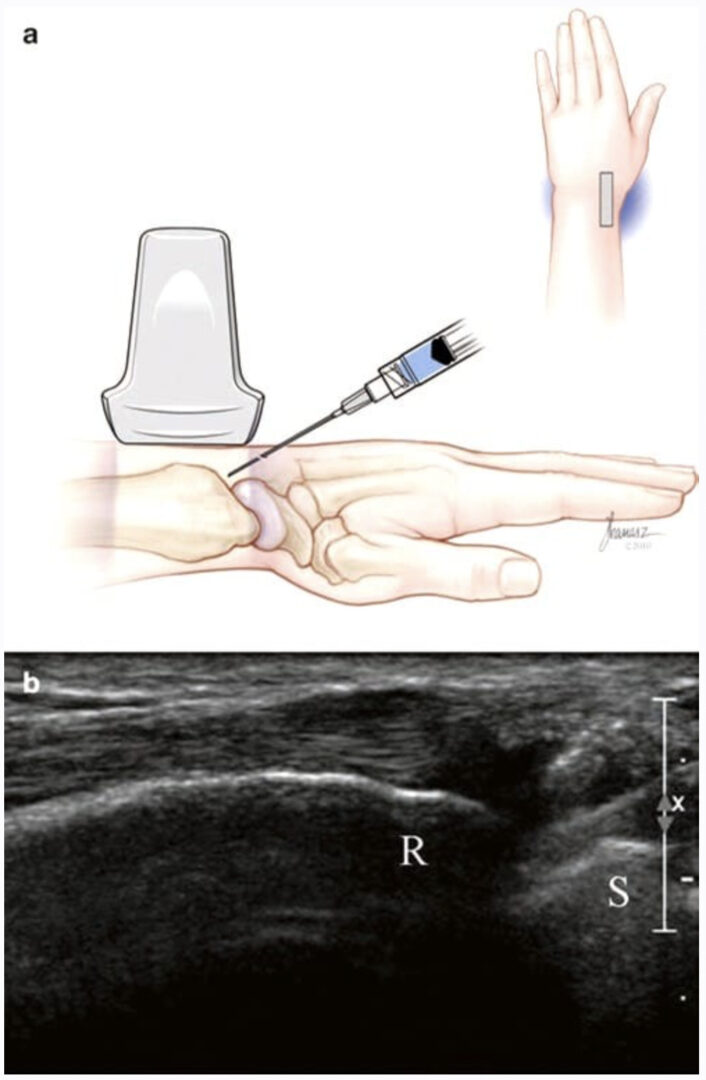
For the long-axis approach to the radiocarpal joint, the patient is seated next to the ultrasound machine facing the physician. The wrist is in pronation and slight volar flexion and resting on a pillow. Lister’s tubercle is identified in the short axis. Next to it on the ulnar side is the extensor pollicis longus (EPL) followed by the extensor digitorum communis (EDC). The interval between EPL and EDC tendons is centered on the screen, and the transducer moved distally until the bony cortex of the radius disappears. Here the transducer is rotated 90° so that the underlying radial-scaphoid joint is seen in the long axis (Fig. 5). A 27-gauge, 32 mm needle is then advanced in the long axis from distally to proximally until the tip of the needle enters the joint.
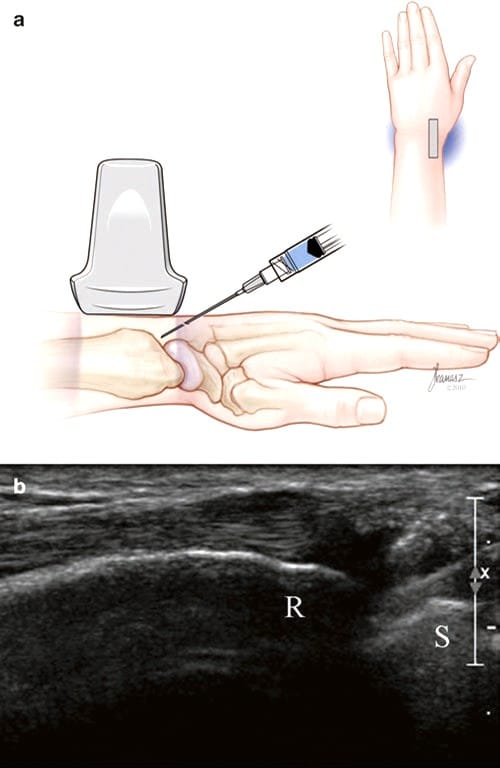
Fig.5 Wrist (radial-scaphoid) joint long-axis injection. (a) Illustration and medical illustrations by Joseph Kanasz, BFA. (b) Long- axis view of the joint and radius (R) and scaphoid (S) and needle seen entering from the right. Injected fluid surrounds the tip of the needle.
For small and superficial joint injections such as the CMC joint of the thumb, a short-axis injection is easiest to perform. The wrist is placed in neutral, between pronation and supination, in slight ulnar deviation for a dorsal approach, and in supination, thumb adduction, and slight ulnar deviation for a volar approach. The joint is centered on the screen, and the distance between the skin and a point within the superficial part of joint is estimated. A 30-gauge, 12.5 or 25 mm needle is inserted in the short axis and directed toward the joint (Fig. 6). When the needle is within the joint, 0.5–1.0 ml of corticosteroid, lidocaine, or viscosupplement is injected. The advantage of the dorsal approach is that it avoids the sensitive skin of the volar aspect of the hand, whereas the volar approach, as described by Umphrey et al. avoids the overlying thumb tendons.
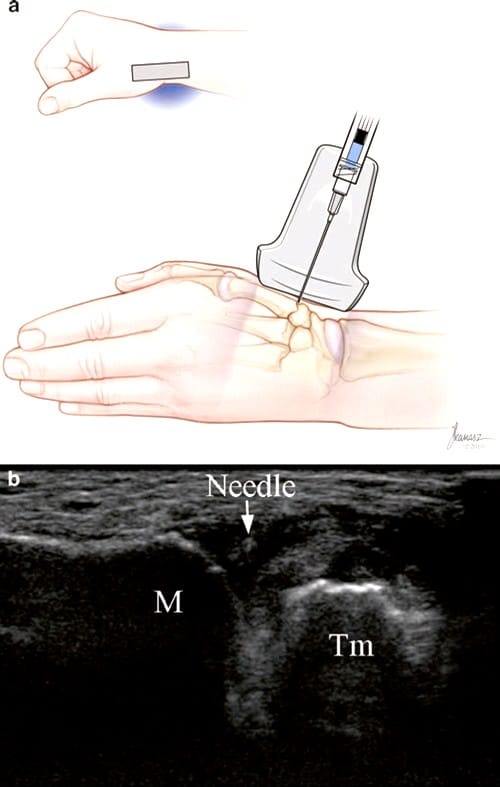
Fig.6 CMC joint of thumb injection (short-axis dorsal approach). (a) Illustration and medical illustrations by Joseph Kanasz, BFA. (b) Short-axis view of the needle (arrow), the proximal metacarpal bone (M), and trapezium (Tm) during injection. The medication is being injected via a 30-gauge needle resulting in high velocity and air bubbles being injected deep into the joint producing a somewhat brighter appearance of the fluid between M and Tm.
10. ANATOMY ULTRASOUND-GUIDED INJECTIONS FOR TENDON DYSFUNCTION
The extensor tendons are divided into six compartments at the dorsal wrist and forearm: E1, abductor pollicis longus (APL) and extensor pollicis brevis (EPB); E2, extensor carpi radialis longus and brevis (ECRL and ECRB); E3, EPL; E4, EDC; E5, extensor digiti minimi (EDM); and E6, extensor carpi ulnaris (ECU). The tendons are prone to friction, overuse, effusions, and degenerative changes. The common extensor tendon of the ECRB, EDC, EDM, and ECU originates from the lateral epicondyle of the humerus. The anatomy of the flexor tendons is discussed in the carpal tunnel section.
11. TENDON DYSFUNCTION
de Quervain’s Tenosynovitis
Fritz de Quervain described stenosing tenosynovitis of the first compartment tendons, the APL and EPB, in 1895. Pain with thumb and wrist motion and tenderness over the radial styloid are present. The incidence is approximately 0.94–6.3 per 1000 person-years, and women, older individuals, and African–Americans are at greater risk. Ultrasound findings include tendon and synovial sheath thickening with peritendinous edematous changes.
Zingas et al. performed blind injections of corticosteroid and radiographic dye in 19 patients with de Quervain’s tenosynovitis. Relief of symptoms occurred in 11 of 16 in which dye was present in E1, in 4 of 5 in which dye was seen within E1 and around both APL and EPB tendons, and in 0 of 3 in which dye did not get into E1. The authors concluded that the optimal resolution of symptoms depends on accurate tendon sheath injections and hypothesized that if an unrecognized septum separates the smaller EPB from the larger APL, injections and surgery may fail.
Avci et al. performed a randomized controlled trial in pregnant and lactating women demonstrating complete relief of pain in nine of nine patients treated with blind corticosteroid injections and in zero of nine using thumb spica splints.
Jeyapalan and Choudhary performed US-guided injections in 17 patients with de Quervain’s tenosynovitis, noting significant resolution of symptoms in the 15 of 16 (94%) patients that were available for follow-up.
Intersection Syndrome
Intersection or oarsman’s syndrome occurs at the intersection of the E1 (APL and EPB) and E2 (ECRL and ECRB) tendon sheaths in the distal forearm. Focal tenderness to palpation confirms the diagnosis. Ultrasound findings may include thickening of the tendon sheaths or the presence of an effusion. Ultrasound-guided corticosteroid injection and avoidance of direct pressure and exacerbating activities can help resolve this problem. A rarer friction syndrome can occur more distally at the intersection of E2 and E3.
Lateral Epicondylitis
Lateral epicondylitis (LE) or tennis elbow has an incidence of 0.4–0.7% among the general population. LE is secondary to overuse, degeneration, lack of regeneration (tendinosis), or micro-tears of the common extensor tendon. The deep fibers of the ECRB portion of the tendon are most often involved. Ultrasound findings include diffuse tendon enlargement, hypoechoic areas, linear and complex tears, intratendinous calcification, and adjacent bone irregularity.
Recent systematic reviews found that corticosteroid injections provide good short-term relief of symptoms but no long-term benefit, whereas physical therapy slightly improves intermediate and long-term outcomes compared to no intervention. Risks of corticosteroids include common extensor tendon and lateral collateral ligament rupture.
Mishra et al. performed the first randomized controlled trial of platelet-rich plasma (PRP) injections for chronic lateral epicondylitis in 20 patients who had failed corticosteroid injections and physical therapy. After 8 weeks there was a 60% improvement in VAS score among the 15 patients in the PRP group, compared to 16% for the 5 patients in the bupivacaine group. At final follow-up, an average of 25.6 months later, there was a 93% improvement in the PRP group.
Recent systematic reviews also concluded that prolotherapy, polidocanol, autologous whole blood, and PRP are all effective for LE with more studies underway. McShane et al. reported good to excellent results in 92% of patients at an average of 22 months following sonographically guided percutaneous needle tenotomy for LE.
Tendon Impingement
Arora et al. reported on a series of 141 patients treated with a fixed-angle open reduction internal fixation (ORIF) palmar plate, noting two ruptures of the FPL tendon, nine cases of flexor tendon tenosynovitis, two EPL ruptures, four cases of extensor tendon synovitis, three CTS, and five with CRPS. Casaletto et al. described seven cases of FPL rupture associated with palmar plate fixation. Adham et al. described four cases of flexor tendon problems after volar plate fixation of distal radius fractures, all of which were associated with close contact of flexor tendons with screws or the distal edge of the plate.
12. ULTRASOUND-GUIDED TECHNIQUE FOR TENDON DYSFUNCTION
US-guided injections for de Quervain’s tenosynovitis are performed as follows: the APL and EPB tendons are identified in the short axis at the base of the thumb and followed proximally to the point of maximum tenderness, usually where they cross the radial styloid. The E1 tendon sheath is the target for injection, but each tendon can be targeted separately if a septum is present or flow does not spread throughout the sheath. After the cleft between tendons is centered on the screen, a short-axis injection is performed using a 27-gauge, 32-mm needle, and 1–2 ml of lidocaine/ corticosteroid (Fig. 7).
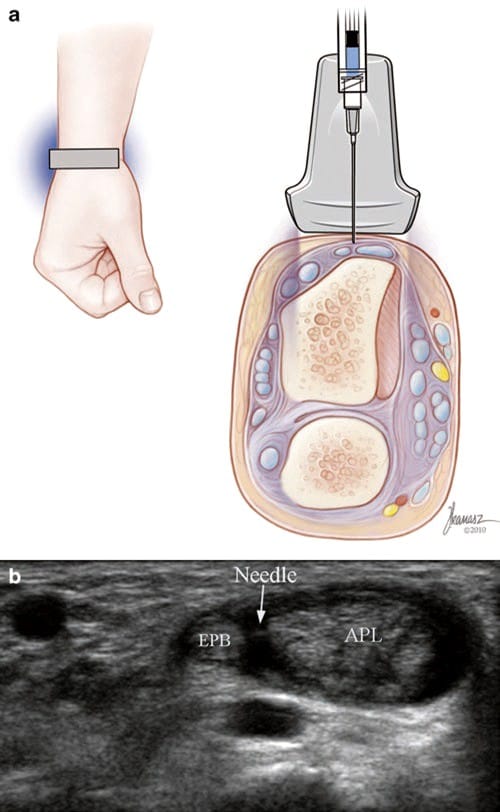
Fig.7 de Quervain’s tenosynovitis (short-axis injection). (a) Illustration and medical illustrations by Joseph Kanasz, BFA. (b) Short- axis view of the tip of the needle (arrow) seen between the APL and EPB tendons.
US-guided injections for intersection syndrome are performed in a similar fashion. The E1 tendons are followed proximally up to the point where they cross the E2 tendons. A short-axis injection can be provided into the E1 tendon sheath between the APL and EPB tendons, followed by advancement of the needle into the space between E1 and E2 where more medication can be injected.
Ultrasonography for lateral epicondylitis is most useful to determine whether the common extensor tendon is swollen, degenerated, and partially or completely torn, factors which are as likely to impact outcome as exact needle placement. Ultrasound guidance can be used in the short or long axis for an injection of PRP into a tear or for the assessment of spread of injectate (Fig. 8).
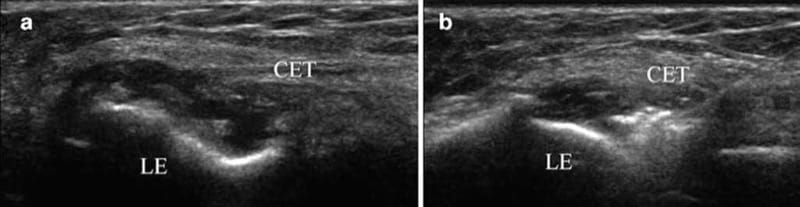
Fig.8 Lateral epicondylitis. (a) Long-axis view showing anechoic fluid between the origin of the common extensor tendon (CET) and the lateral epicondyle (LE) indicating a tear. (b) Long-axis view with needle showing PRP being injected into the tear.
US-guided injections for tendon impingement can be performed following the use of dynamic imaging to determine which tendon is being impinged and where. An injection of local anesthetic only is provided because corticosteroids increase the risk of tendon rupture. When the source of pain is identified, a decision can be made whether to remove the hardware. The injection technique for impingement of a tendon such as the FPL is similar to that for CTS. A short- or long-axis approach is used, but the needle is advanced beyond the superficial row of tendons so that the tip is positioned between the FPL and fixation plate or screw. At that point, 0.5–1.0 ml of lidocaine 4% or bupivacaine 0.75% is injected followed by the assessment of pain and function (Fig. 9).
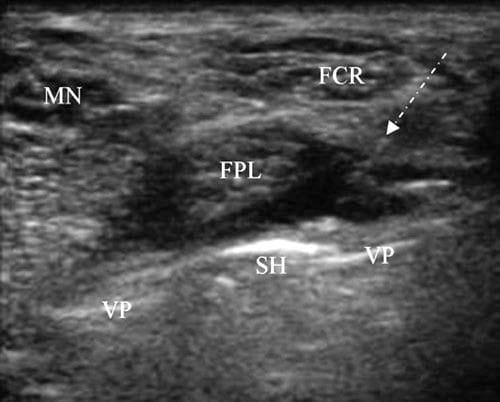
Fig.9 FPL tendon impingement. Short-axis view of the distal radius with a volar fixation plate (VP) and protruding screw head (SH) adjacent to the FPL. The image was taken during a diagnostic injection. The FPL tendon is being displaced away from the SH by local anesthetic injected via a long-axis approach. The needle is seen as a series of dots below the arrow and is difficult to see because of its high angle.
13. ANATOMY ULTRASOUND-GUIDED ELBOW INJECTIONS
The elbow is a compound joint formed by the articulations of three bones including the humerus, radius, and ulna. The ulno-humeral articulation approximates a hinge joint, whereas the radioulnar and radio-humeral articulations allow for axial rotation. The joint capsule envelopes the entire elbow joint and is taut in elbow extension and lax in elbow flexion. It contains three fat pads, two of which are located in the capitellar and trochlear fossa and the third in the olecranon fossa. When an elbow joint effusion is present, the fat pads are elevated, resulting in the radiographic signs of visible posterior and elevated anterior fat pads.
Numerous bursae are found around the elbow including the cubital and olecranon bursae. The cubital bursae include the bicipito-radial bursa and the interosseous bursa. The cubital bursa is located between the distal biceps tendon and the radial tuberosity and decreases friction during forearm pronation. Cubital bursitis is rare and causes pain and swelling in the antecubital fossa. Three bursae are found posteriorly including the superficial olecranon bursa that is located in the subcutaneous tissue posterior to the olecranon. This bursa is commonly inflamed following direct injury or repetitive trauma or with inflammatory disorders.
Knowledge of peripheral nerve anatomy around the elbow is important when performing interventional procedures in this area. The ulnar nerve is located medially between the olecranon process and medial epicondyle, and the radial nerve is located laterally under the brachioradialis muscle, where it bifurcates into deep and superficial branches. The deep branch of the radial nerve runs between the two heads of the supinator, and the superficial branch runs under the brachioradialis muscle on its way to the dorsal radial aspect of the hand. The median nerve lies anteriorly, superficial to the brachialis muscle and medial to the brachial artery.
14. LITERATURE REVIEW ON ULTRASOUND-GUIDED ELBOW INJECTIONS
Ultrasound-guided elbow joint injections are commonly performed for diagnosis and treatment of pain resulting from osteoarthritis, rheumatoid arthritis, crystal arthropathies, and infection. Ultrasound can be a valuable tool to the physician treating elbow pain since physical examination and blind aspiration often fail to reveal the presence of an effusion.
Louis et al. and Bruyn et al. described similar approaches with the elbow either flexed across the chest or protruding behind the back with the hand resting on a flat surface. The transducer is aligned with the long axis of the upper arm and moved laterally until just out of view of the triceps tendon. The needle is inserted using a long-axis approach. The median, radial, and ulnar nerves are not at risk for injury with this approach, and the key anatomical landmarks include the concave olecranon fossa of the humerus, the posterior fat pad, and the olecranon.
15. ULTRASOUND-GUIDED ELBOW INJECTION TECHNIQUE
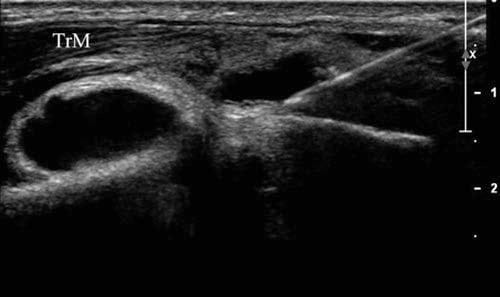
The patient is seated facing away from the physician with a pillow doubled-up in the lap, the hand resting on the pillow, and the elbow bent. A long-axis view of the olecranon and triceps tendon is obtained (Fig. 10a). While maintaining the lower end of the transducer on the olecranon, the upper end is rotated 30° clockwise for the right or 30° counter clockwise for the left elbow. As the transducer is rotated, the convex surface of the lateral trochlea of the distal humerus with its thin layer of hypoechoic cartilage emerges into view. The joint space is the small notch between the olecranon and trochlea (Fig. 10c).
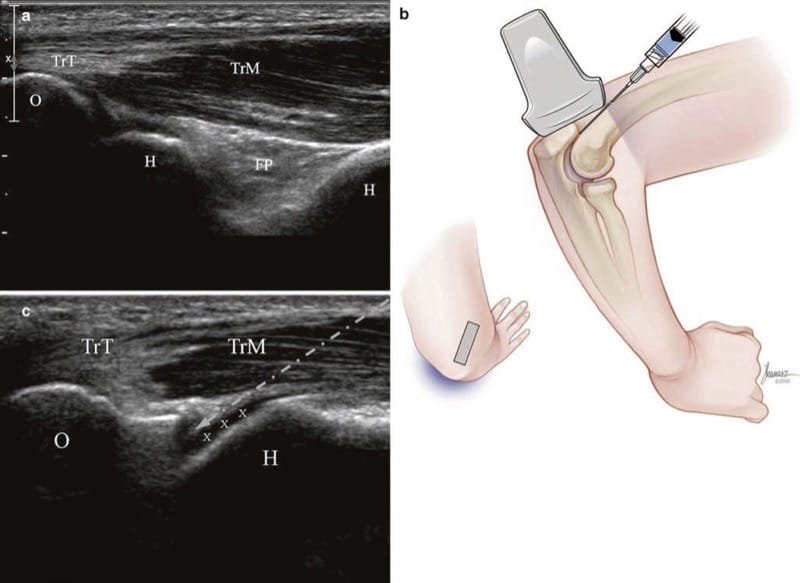
Fig.10 Elbow (long-axis injection). (a) Initial long-axis view of the triceps tendon (TrT), muscle (TrM), olecranon (O), humerus (H), hyaline cartilage (x), and posterior fat pad (FP). (b) Illustration of posi-tion after rotating the upper end of the transducer 30° laterally. Medical illustrations by Joseph Kanasz, BFA. (c) Ultrasound image (b) showing triceps tendon (TrT), muscle (TrM), and needle trajectory (arrow) which passes through the muscle and avoids the tendon.
One must be careful not to rotate too far laterally – if the hypoechoic layer of cartilage is not seen, the bony surface seen superior to the olecranon may be the posterior lateral epicondyle. The transducer is then moved inferiorly to minimize the distance the needle needs to travel to the joint space. As usual, the thinnest possible needle is used and is inserted in the long axis from superiorly to inferiorly (Fig. 10b). If an aspiration needs to be performed (Fig. 11), the needle is withdrawn while anesthetizing its track, and a larger-gauge needle is inserted along its path.
Clinical updates
- Palmbergen et al. (The Lancet, 2025) in the DISTRICTS multicentre RCT (n=934) found that initiating treatment for carpal tunnel syndrome with surgery resulted in higher 18-month recovery than starting with corticosteroid injection (61% vs 45%; RR 1.36, 95% CI 1.19–1.56), with a shorter median time to recovery (9 vs 18 months). Although injections provided greater symptom relief at 6 weeks, from 3 months onward, surgery showed superior symptom scores and function, with similar overall adverse event rates (86% vs 85%) and only one surgery-related hospitalization.
Palmbergen WAC, Beekman R, Heeren AM, et al. Surgery versus corticosteroid injection for carpal tunnel syndrome (DISTRICTS): an open-label, multicentre, randomised controlled trial. Lancet. 2025;405(10495):2153-2163.
- Chan et al. (HAND, 2025) systematically reviewed 20 studies (n=3688) with ≥1-year follow-up and reported that 41.6% of patients ultimately required carpal tunnel release after initial corticosteroid injection, 29% underwent reinjection, and no major complications were identified (3.0% minor events), with median time to surgery ranging 128–446 days. Long-term functional outcomes were heterogeneous, but injections frequently delayed, rather than prevented, subsequent surgery.
Chan PYW, Santana A, Alter T, Shiffer M, Kalahasti S, Katt BM. Long-term Efficacy of Corticosteroid Injection for Carpal Tunnel Syndrome: A Systematic Review. Hand (N Y). 2025;20(3):463-473.