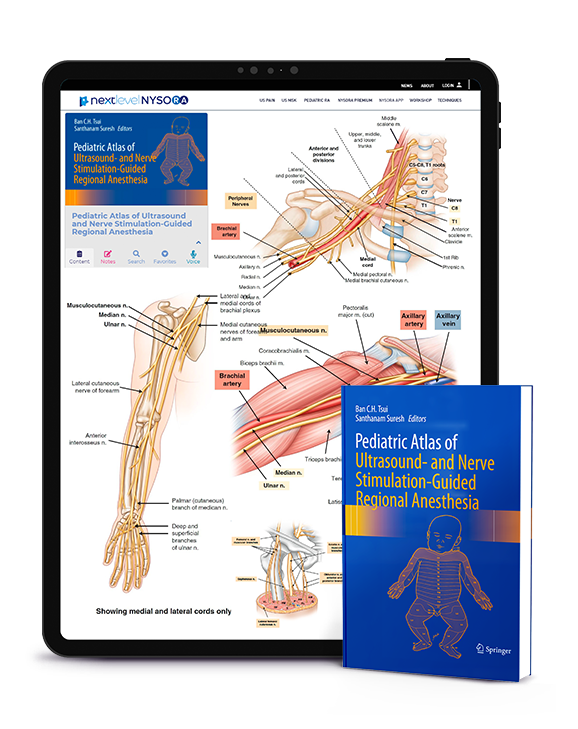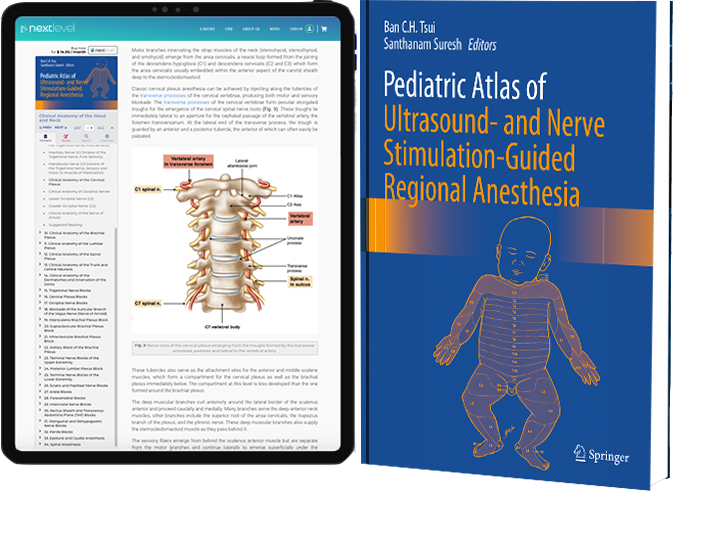
Pediatric Regional Anesthesia, Review
Introduction – Pediatric Regional Anesthesia Over the past 30 years, pediatric regional anesthesia has come of age. There is now a large and rapidly growing body of information regarding the impact of development on pain responses, local anesthetic pharmacology, and the adaptation of a full range of regional anesthetic techniques for infants and children. There is also a quickly expanding literature on safety and efficacy from registries and clinical trials. Thirty years ago, in most pediatric centers worldwide, regional anesthesia was used for only a small fraction of surgeries. Today, it is an essential part of pediatric anesthetic and analgesic management throughout the world. For children in tertiary centers in highly developed countries, regional anesthesia is recognized as an essential component of multimodal analgesic regimens that seek to provide pain relief with movement; diminish opioid use, with a corresponding reduction in opioid side-effects; and facilitate early mobilization, early enteral feeding, and early hospital discharge. Some preliminary studies in infant humans and infant animals suggest that regional anesthesia may have an impact on preventing prolonged changes in central nervous system responses to surgical trauma. In lower resource settings, pediatric regional anesthesia is more often used as a primary anesthetic approach, based on considerations of cost, safety, and reduced need for postoperative intensive care. In the face of ongoing controversy over the impact of general anesthetics on the developing brain, regional anesthesia has a growing role for neonates, infants, and toddlers as an approach to limiting general anesthetic dosing and overall exposure. Overview Ban Tsui and Santhanam Suresh have been pioneers in this effort, and it is fitting that they are co-editing this wonderful textbook. Both editors have made fundamental innovations in the field over the past 20 years, and both continue to innovate and to mentor to a new generation of