Learning objectives
- Describe the contributing factors to high spinal anesthesia
- Apply preventative measures for high spinal anesthesia
- Describe the symptoms of high spinal anesthesia
- Manage cases of high spinal anesthesia
Definition & mechanisms
- High spinal anesthesia is a complication of central neuraxial techniques that include spinal and epidural anesthesia
- It is defined as a spread of local anesthetic affecting the spinal nerves above T4
- The effects are of variable severity depending on the maximum level that is involved but can include cardiovascular and/or respiratory compromise
- In total spinal anesthesia, there is an intracranial spread of local anesthetic resulting in loss of consciousness
Contributing factors
- Local anesthetic dose
- Positioning of patient
- Pre-existing epidural block
- Unrecognized dural puncture and intrathecal injection
- Accidental subdural block
- Accidental intradural space
Prevention
- Epidural analgesia/anesthesia:
-
- Use low concentrations of local anesthetic for labor analgesia
- Prior to top-up:
- Assess block (to guide top-up dosage)
- Aspirate the epidural catheter with a 2 mL syringe to rule out intrathecal or intravenous placement
- Consider giving large volumes of local anesthetic in divided doses (clinical urgency may preclude this)
- Spinal anesthesia:
-
- Consider the level (and therefore local anesthetic dose) required for surgery
- Patient position: block height can be manipulated for up to 30 min when using hyperbaric (“heavy”) anesthetics – if using head down position to establish the block, remember to remove it as soon as possible
- Patient characteristics: consider dose reduction in short or morbidly obese patients
- Technique:
- Consider the effects of the speed of injection
- Avoid excessive barbotage
- If performing a spinal following an epidural, a dose reduction may be necessary depending on the existing level of block (reductions to 1-1.5 mL of local anesthetic have been suggested following a failed epidural top-up); there is no clear consensus on this
- Epidural and spinal anesthesia:
-
- Don’t inject during a contraction/cough/Valsalva maneuver as this can increase the cephalad spread of local anesthetic
- The use of the Oxford wedge is recommended to prevent the cephalad spread of local anesthetic (and to optimize airway positioning in the event of requiring general anesthesia)
Symptoms
| Spinal level | Area(s) affected | Symptoms |
|---|---|---|
| T1-T4 | Cardiac sympathetic fibers blocked | Hypotension Bradycardia |
| C6-C8 | Hands and arms | Paresthesia or numbness in hands/arms Weakness of hands/arms Shortness of breath (accessory respiratory muscles affected) |
| C3-5 | Diaphragm and shoulders | Shoulder weakness – respiratory compromise imminent Hypoventilation and/or desaturation Respiratory arrest |
| Intracranial spread | Brain stem | Slurred speech Sedation Loss of consciousness |
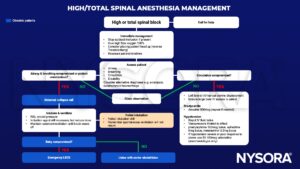
Management
Suggested reading
- Sivanandan S., Surendran A. (2019) Management of total spinal block in obstetrics. Update in Anaesthesia, 34: 22-25.
- Reeve J. (2017) NHS Foundation trust clinical guideline: High Regional Block (including Total Spinal Anaesthesia).
We would love to hear from you. If you should detect any errors, email us at customerservice@nysora.com